 |
 |
| Plant Pathol J > Volume 31(4); 2015 > Article |
Abstract
Citrus canker caused by Xanthomonas citri subsp. citri (Xcc) is one of the most important bacterial diseases of citrus. Because citrus canker is not found in many countries including European Union and Australia, Xcc is strictly regulated in order to prevent its spread. In this study, the effects of X-irradiation on Xcc growth either in the suspension or on the surface of citrus fruits were investigated. The suspension containing 1×107 cfu/ml of Xcc was irradiated with different absorbed doses of X-irradiation ranging from 50 to 400 Gy. The results showed that Xcc was fully dead at 400 Gy of X-irradiation. To determine the effect of X-irradiation on quarantine, the Xcc-inoculated citrus fruits were irradiated with different X-ray doses at which Xcc was completely inhibited by an irradiation dose of 250 Gy. The D10 value for Xcc on citrus fruits was found to be 97 Gy, indicating the possibility of direct application on citrus quarantine without any side sterilizer. Beside, presence of Xcc on the surface of asymptomatic citrus fruits obtained from citrus canker-infected orchards was noted. It indicated that the exporting citrus fruits need any treatment so that Xcc on the citrus fruits should be completely eliminated. Based on these results, ionizing radiation can be considered as an alternative method of eradicating Xcc for export of citrus fruits.
Citrus canker caused by Xanthomonas citri subsp. citri (Xcc) is one of the most serious bacterial diseases of many commercial citrus varieties, resulting in significant economic losses worldwide (Schaad et al., 2006). Xcc has been recently reclassified from Xanthomonas axonopodis pv. ctiri (Schaad et al., 2006; Vauterin et al., 1995). The pathogen is spread by rain splash and enters its host plant through stomata and wounds. They cause distinctive necrotic raised lesions surrounded by oily, water-soaked margins and yellow chlorotic rings on leaves, stems, and fruits (Graham et al., 2004; Schubert et al., 2001). Severe infection causes defoliation, dieback and fruit drop, which have serious economic consequences (Gottwald et al., 2002).
The disease first appeared in Southeast Asia and India, but it is now present in more than 30 countries such as Japan, South and Central Africa, South America, and USA (Das, 2003; Gottwald et al., 2001; Mohammadi et al., 2001; Stall and Civerolo, 1991). In some locations, efforts to eradicate Xcc have failed and continued active eradication campaigns are being performed (Schubert et al., 2001). In contrast, Xcc has not been detected in many countries such as European Union or Australia (Del Campo et al., 2009), where Xcc was considered as a quarantine organism and strict regulatory programs were implemented to prevent the spread of citrus canker disease. Consequently, only the disinfected and asymptomatic citrus fruits are allowed to be exported (Golmohammadi et al., 2007). However, because even asymptomatic citrus fruits were suspected of harboring Xcc, a completely eradication system is very essential for export.
As an eradication measure, methyl bromide (MeBr) has been used for effective control of nematodes, fungi, and insects on more than 100 crops in the world. However, MeBr is identified as an ozone-depleting substance and it may cause serious problems to human health and environment (Bell et al., 1996; Penner, 1999; Watson et al., 1992). Under the Montreal Protocol, developed countries were required to completely phase out MeBr use by 2015 (Fields and White, 2002; Gareau, 2010; Osteen, 2003). Therefore, it is necessary to develop an eco-friendly quarantine treatment for replacing the use of MeBr fumigation.
Recently, the use of ionizing radiation as a promising phytosanitary treatment is increasing all over the world (Hallman, 2011). Thousands of researches have reported that irradiation enhances the quality, safety, and marketability of fruits and vegetables (Abu-Tarboush et al., 1997; Jeong, 2014; Mostafavi et al., 2013). Irradiation does not leave any residues and does not make food harmful for consumers. Also, comparisons of fruit quality against traditional MeBr fumigation showed that irradiation provided better overall quality than MeBr fumigation (Drake and Neven, 1997).
The sterilization mechanisms for irradiation of living cells are well known (Aquino, 2012). Irradiation causes injury to plant cells, insects or microorganisms on the produce due to gamma irradiation, e-beams, or X-irradiation (Kader, 1986). Ionizing radiation exerts its effect directly by breaking the chromosomal DNA and indirectly by producing oxygen radicals which can disrupt membranes and interfere with the DNA (Elgazzar and Kazem, 2015; Niemira, 2003). Irradiation treatment up to 10 kGy has been considered a safe and effective technology since 1981 by several international food organizations (World Health Organization, 1981). Its application can eliminate the pathogenic organisms without causing any toxicological hazard and loss of quality of fruits (World Health Organization, 1981; Youssef, 1994).
It has been reported that ionizing radiation of citrus fruits prevents decay and extends the shelf-life (Mahmoud et al., 2010). However, the effect of X-irradiation on Xcc, which is important for citrus quarantine, has not been reported. The objective of this study was to determine the X-irradiation sensitivity of Xcc so that the quarantine system using X-irradiation can be applied to the citrus fruits being exported. For this purpose, we determined the accurate dose of X-ray which completely eliminates Xcc either in the suspension or on the surface of Satsuma mandarin fruits.
The bacterium was isolated from leaves which were naturally infected with the citrus canker pathogen at a citrus orchard in Topyung, Jeju. The experiments were carried out in a clean bench to prevent secondary contamination. Edges of a citrus canker lesion on a leaf were cut into small pieces (5×5 mm) with a blade and were surface-sterilized with 1% sodium hypochlorite solution (NaClO) for 30 sec, rinsed in sterile distilled water three times each for 1 min. Then, the samples were sterilized again with 70% ethanol for 30 sec and likewise rinsed. The samples were divided into four parts, placed in 5% of peptone solution, and shaken at room temperature for 2 h at 70 rpm by a shaker (CR300, Finemould Precision Ind. Co., Korea). An aliquot of 100 μl of the solution was inoculated into tryptic soy agar medium (TSA: Becton, Dickinson and company, France) at 28°C for 48 h. Stock cultures were maintained on 20% glycerol at −80°C.
For identification of the bacteria, the genomic DNA was extracted from the bacteria using a genomic DNA extraction kit (Dneasy Blood & Tissue Kit 56, QIAGEN™, Germany). PCR amplification of the internal transcribed spacer region (ITS) of 16S/23S ribosome was carried out using primer 2 (5′-CACGGGTGCAAAAAATCT-3′) and primer 3 (5′-TGGTGTCGTCGCTTGTAT-3′) (Hartung et al., 1993). PCR reactions were performed in volume of 40 μl containing final concentrations of 10× PCR buffer (iNtRON Biotechnology Inc., Seoul, Korea), 2.5 mM dNTP, 0.5 μM each primer (2 and 3), 10 ng of template DNA, and 5.0 U of Taq DNA polymerase (iNtRON). PCR amplifications were run in Thermal Cycler TP600 (TaKaRa, Japan) with an initial denaturation step at 94°C for 3 min; followed by 36 cycles of denaturation at 94°C for 45 s, annealing at 60°C for 45 s and elongation at 72°C for 45 s; plus a final extension at 72°C for 5 min. The PCR products were visualized on 1% agarose gels stained with 0.01% ethidium bromide under ultraviolet light. Nucleotide sequences of the PCR products were identified at a commercial analysis service (Macrogen Inc., Seoul, Korea). The sequences were compared to those in the GeneBank database using the NCBI BLAST program.
A semi-selective medium (SSM) was used to detect Xcc selectively (Dezordi et al., 2009). SSM was prepared by adding the following compounds: peptone 5 g, beef extract 3 g, sucrose 5 g, soluble starch 10 g, agar 15 g, CaCl2 0.25 g, Tween 80 10 ml, 1% crystal violet solution 150 μl in 1 l total volume with distilled water. And cephalexin 50 mg, methyl thiophanate 10 mg, and chlorothalonil 10 mg were added after autoclaving the culture medium.
One hundred microliters of the stock culture of Xcc was inoculated into tryptic soy broth medium (TSB: Becton, Dickinson and company, France) and incubated at 28°C for 48 h at 120 rpm using a shaking incubator (HB-201SL, Hanbaek Scientific Co., Korea). An aliquot of 10 ml of the suspension was transferred to a conical test tube and the concentration was adjusted to 1×107 cfu/ml by a UV-visible spectrophotometer (Optizen POP, Mecasys co., Korea) at a wavelength of 600 nm. In 3 independent trials, the suspensions of Xcc were irradiated using a X-irradiator (10 MeV Linear Electron accelerator, MB10/8-635, Mevex, Canada). All 3 tubes were exposed to different absorbed doses of 0, 50, 100, 150, 200, 250, 300, 350, and 400 Gy at a dose rate of 0.7 kGy/h. Alanine pellets were used for dosimetry (ES 200-2106/E2044562, Bruker Biospin, Germany). Following treatment, both irradiated and non-irradiated suspensions of Xcc were serially diluted with sterile water. After dilution, 100 μl of each dilute was spread on SSM in triplicate. After incubation at 28°C for 3 days, the number of colonies was counted.
To assess the strength of the doses needed to eradicate Xcc on the surface of citrus fruits, the following experiments were carried out. Satsuma mandarin fruits were purchased from a local market in Jeju. The fruits measuring about 60 mm of diameter were selected for the experiments. Fruits were washed under running water, and then dried and cleaned to remove any dirt and microorganisms. The fruit samples were sprayed with Xcc suspension at a concentration of 1×107 cfu/ml and 0.01% Tween 20 evenly until dew moist. After the fruits were completely dried at room temperature, the citrus fruits were exposed to X-irradiation at absorbed doses of 0, 30, 50, 100, 150, 200, 250, and 300 Gy in the same way as the Xcc suspension mentioned above. After irradiation, the fruits were placed in a beaker which was filled with sterile water until the fruits were submerged and shaken at 100 rpm at 28°C for 2 h. Then 100 μl of the washing solution was prepared by plating on the SSM and incubated at 28°C for 3 days.
The radiation sensitivity of Xcc was measured by the D10 value which is defined as the radiation dose required to reduce the microbial population by 90%. After incubating the suspension or the washing solution of Xcc at 28°C for 3 days, the colony forming units (cfu) of Xcc were counted. The D10 value was determined by the survival curve method. The number of cfu per sample was plotted as log10 against radiation doses. Each experiment was carried out three times and the D10 value was generated. The slopes of the individual survivor curves were calculated with linear regression using a graphic program of Microsoft Excel.
Asymptomatic Satsuma mandarin fruits were obtained from citrus canker infected orchards. The microorganisms on the surface of the fruits were isolated on the SSM using the same methods by which Xcc was isolated from the X-irradiated fruits. Among various growing epiphytic bacteria, 6 colonies per plate were randomly selected, spread on TSA media, and incubated at 28°C for 3 days.
Identification of the isolated epiphytic bacteria was performed by colony polymerase chain reaction (PCR). Colony PCR is a very easy to perform, effective technique that allows rapid amplification of DNA fragments and screening of a large number of bacterial colonies (Cao et al., 2009). For direct PCR amplification, the PCR reaction mixtures contained 1× PCR buffer, 1.5 mM of MgCl2, 1.0 unit of Taq DNA polymerase (Invitrogen Corp, San Diego, CA, USA), 250 μM of each dNTP, 3.6 mM of each primer Xac01 (5′-CGC CAT CCC CAC CAC CAC CAC GAC-3′) and Xac02 (5′-AAC CGC TCA ATG CCA TCC ACT TCA-3′) (Coletta-Filho et al., 2006), and distilled water up to a total volume of 20 μl. All isolated bacteria stored in the Eppendorf tube were centrifuged at 12,000 rpm for 5 min. The bacterial pellet was diluted with 1 ml of sterile water, of which 1 μl was directly added to the PCR tubes as a DNA template. PCR amplification and analysis of the nucleotide sequences were carried out by the same methods as those used for identification of Xcc mentioned above.
In order to determine the effect of X-irradiation on Xcc, the Xcc suspension was irradiated with absorbed doses ranging from 0 to 400 Gy of X-ray. The number of colonies of Xcc was significantly decreased with increasing radiation doses of X-ray and Xcc was completely inactivated at 400 Gy, which was the total lethal dose (Fig. 1, Table 1). In addition, to determine the dose of X-irradiation for application in quarantine, Satsuma mandarin fruits inoculated with Xcc were irradiated with various doses ranging from 0 to 300 Gy. Similar to that in the Xcc suspension, in the washing solution from the inoculated fruits, the number of surviving Xcc was decreased with the increasing level of X-irradiation (Table 2). However, the total lethal dose was >250 Gy for the surface of citrus fruits, which was lower than that for the Xcc suspension (Tables 1 and 2). Probably, it need stronger X-irradiation to sterilize Xcc in the suspension which contained higher concentration in of Xcc than that on the surface of the citrus fruit.
It is well known that ionizing radiation results in inactivation of microorganisms including insects, fungi, bacteria and viruses (Aquino, 2012). Some bacterial populations such as those of Escherichia coli, Salmonellae, and Campylobacter jejuni were reduced with increasing doses of γ-irradiation. They were completely inactivated at 2.5 kGy, the total lethal dose (Clavero et al., 1994; Lee et al., 2006). In our previous study, Xcc was very sensitive to γ-irradiation and its total lethal dose was between 300 and 400 Gy (Kim et al., 2014). This dose was lower than that for E. coli which is known to be a microorganism sensitive to radiation (Mayer-Miebach et al., 2005). Also, in this study it was demonstrated that Xcc was very sensitive to X-irradiation, as it is to γ-irradiation (Table 1). Additionally in case of mold strains, the lethal doses for Rhizopus stolonifer, Botrytis cinerea, Botrytis elliptica, and Aspergillus flavus were 2.8 kGy, 4 kGy, 2 kGy, and 10 kGy, respectively (Aquino et al., 2005; Jeong et al., 2014; Kim and Yun, 2014; Yoon et al., 2014), which were higher compared with that for Xcc. This high sensitivity of Xcc to ionizing radiation may be useful for its application in agricultural production in which elimination of Xcc is required for export to the Xcc- free countries.
To describe the effect of X-irradiation on Xcc more clearly, the D10 values for both Xcc suspension and washing solution were calculated by the survival curves. The number of surviving Xcc after X-irradiation was expressed as log10 cfu/ml and plotted against the irradiation dose (Fig. 2). The D10 value for Xcc in the suspension was 69 Gy (Fig. 2A), whereas the D10 value for Xcc in the washing solution was 97 Gy (Fig. 2B). The higher D10 value for Xcc on the surface of Satsuma mandarin may be caused by the lower water content rather than those in the suspension. Normally, microorganisms are resistance to irradiation in dry condition in which free radicals forming from H2O molecules is lower than in moisture condition (Aquino, 2012). The slope of the graph indicated that inactivation of Xcc was directly correlated to the increasing X-irradiation dose.
In this study, it was observed that the D10 value for Xcc was relatively lower than that of other microorganisms (Fig. 2). For example, the D10 value for E. coli, Salmonella spp., and Yersinia enterocolitica was 360, 610, and 150 Gy, respectively (Sommers and Boyd, 2006). Also, in the case of mold strains such as A. flavus, B.cinerea, and Curvularia geniculata, their D10 values, i.e. between 1.0 to 2.5 kGy, were higher than that of Xcc (Maity et al., 2011; Yoon et al. 2014). Normally, the D10 values for viruses are higher than those of bacterial strains or fungi, which were calculated to range from 3 to 5 kGy (Grieb et al., 2005). These differences in irradiation sensitivity of pathogens may be due to their chemical and physical structure or their ability to recover from the radiation injury (Aquino, 2012; Farkas, 2006).
On the other hand, it should be assessed whether the quality of citrus fruits can be negatively affected by irradiation. Application of ionizing radiation is increasingly being accepted for reducing the risk of pathogenic bacteria in foods. Irradiation dose up to 10 kGy is considered safe and does not affect the nutritional quality of foods (World Health Organization, 1981; Youssef, 1994). Irradiation at a dose of 1 to 3 kGy can be used to enhance the microbial safety without a significant loss in the quality attributes of Chinese cabbage (Ahn et al., 2005). Moreover, there was no significant increase in the mutagenicity between irradiated and non-irradiated fruits (Van Kooij et al., 1978). In the case of citrus fruits, no differences in quality of fruits were detected after an X-irradiation dose of even 300 Gy (Han et al., 2014), which is also the recommended dose of ionizing radiation for eradication of Xcc. Therefore, a low D10 value of 300 Gy for Xcc may be a prerequisite for the application of irradiation as a phytosanitary treatment to eradicate Xcc.
To verify the presence of Xcc on the surface of asymptomatic Satsuma mandarin fruits obtained from citrus canker-infected orchards, microorganisms from the surface of the fruits were identified by colony PCR. Colony PCR is a method for amplifying DNA fragments by PCR using the single colony of organisms without isolating pure DNA. It is a rapid, reliable, and highly accurate detection method, and it is suitable for screening a large number of environmental isolates (Sheu et al., 2000).
Yellow colonies were randomly selected to evaluate the presence of Xcc on asymptomatic Satsuma mandarin fruits (data not shown). Each isolated colony was transferred onto TSA media and used as a DNA template. Total 4 among the 54 selected colonies were identified as Xcc by colony PCR using primers Xac01 and Xac02 (Fig. 3). The results proved the presence of Xcc on asymptomatic citrus fruits, which has the potential ability to spread to susceptible citrus trees causing citrus canker disease. Similar results were reported, which suggested that amplification was observed in asymptomatic leaves of ‘Natal’, ‘Pera’, and ‘Valencia’ sweet orange varieties in Xcc-infected areas (Coletta-Filho et al., 2006). Also, Xcc was detected in 67 out of the 90 samples from leaves with and without symptoms, which were collected from canker-infected pomelo orchards (Kositcharoenkul et al., 2011). Therefore, there is a need for treatment of the citrus fruits being exported so that Xcc bacteria present on the citrus fruits are completely eradicated. Based on these results, X-irradiation as a sterilization strategy is the best way to eliminate all Xcc bacteria present even on asymptomatic citrus fruits.
Acknowledgments
This research was supported by 2015 Export Promotion Technology Development Program, Ministry of Agriculture, Food and Rural Affairs.
Fig. 1
Suppression of colony formation in the suspension of Xcc on semi-selective medium. The suspension of Xcc was exposed to 0 (A), 100 (B), 150 (C), 200 (D), 250 (E), 300 (F), 350 (G), and 400 (H) Gy of X-irradiation. The plates were coated with 100 μl of the bacterial suspension and incubated at 28°C for 3 days. The concentration of the Xcc suspension was 1×107 cfu/ml.
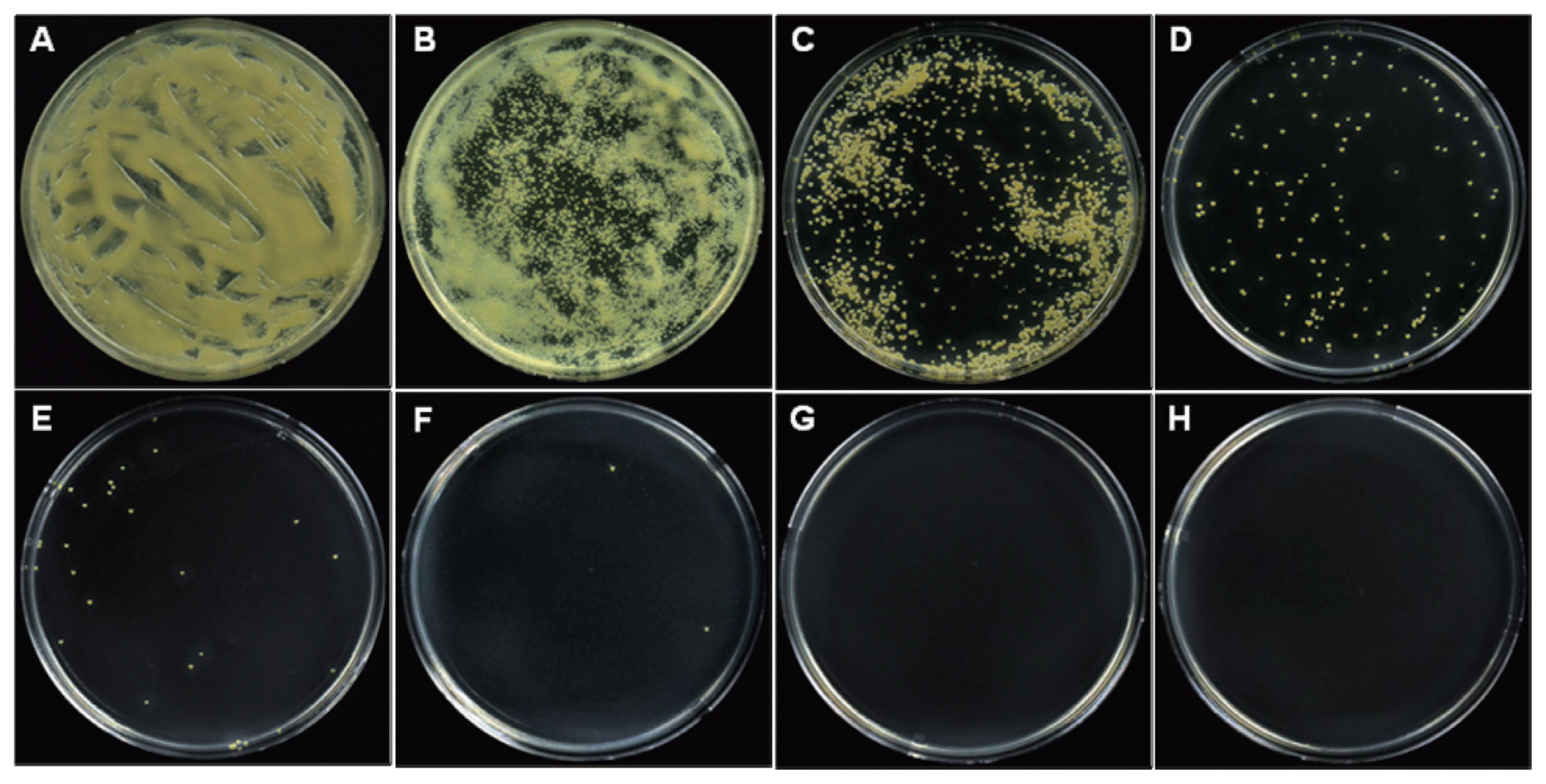
Fig. 2
Survival curves of Xcc in the suspension (A) and in the washing solution from Satsuma mandarin fruits inoculated with Xcc (B) after X-irradiation with different doses. The vertical bars indicate the standard deviation of three separate replications of each experiment.
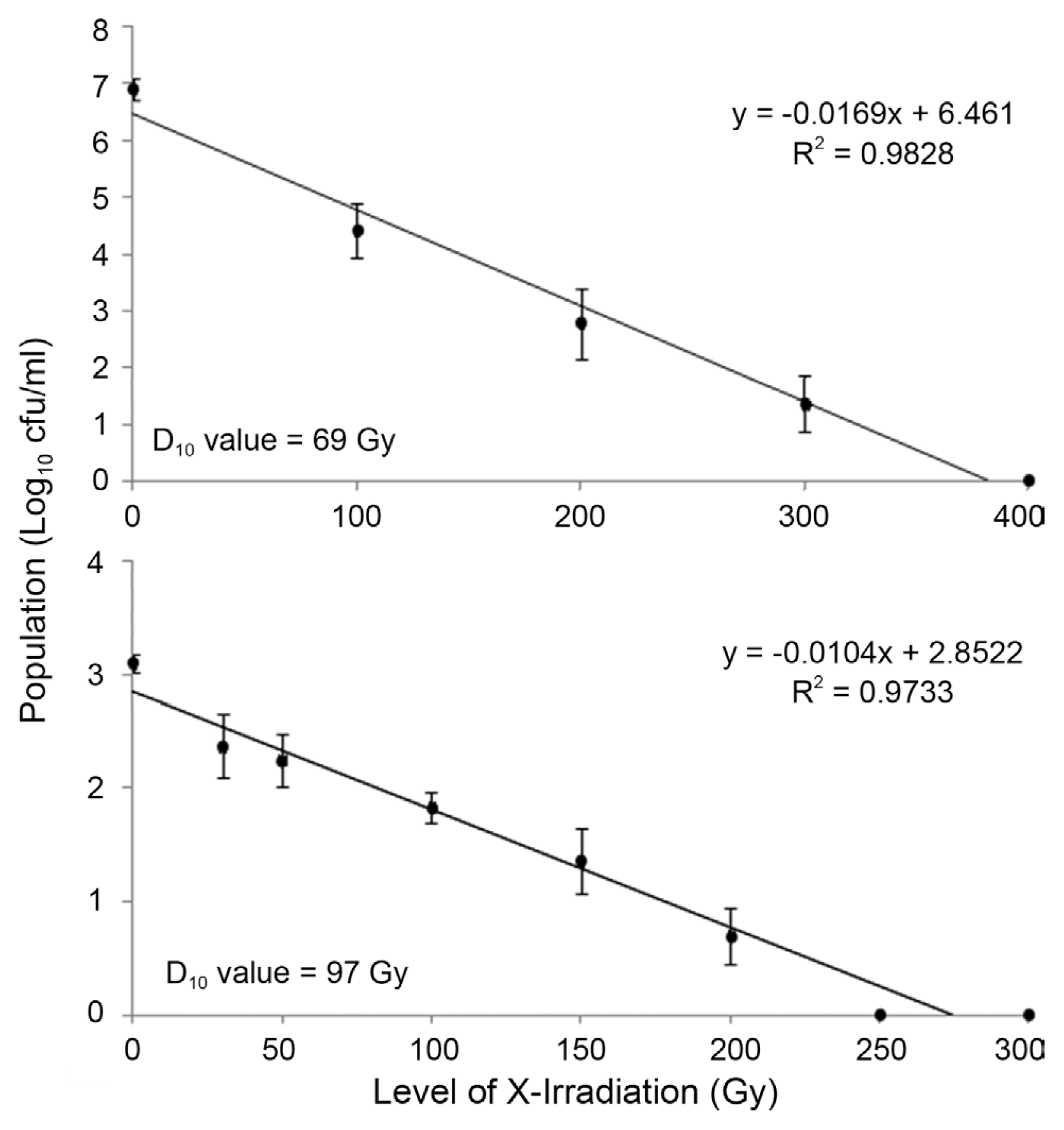
Fig. 3
Detection of Xcc in the washing solution from asymptomatic Satsuma mandarin fruits by colony PCR. Lanes: 1-kb DNA ladder (BIOFACT, Co. Ltd.) (M), epiphytic bacteria from asymptomatic Satsuma mandarin fruits (1-9), Xcc (10). Arrow indicates the Xcc specific fragment.
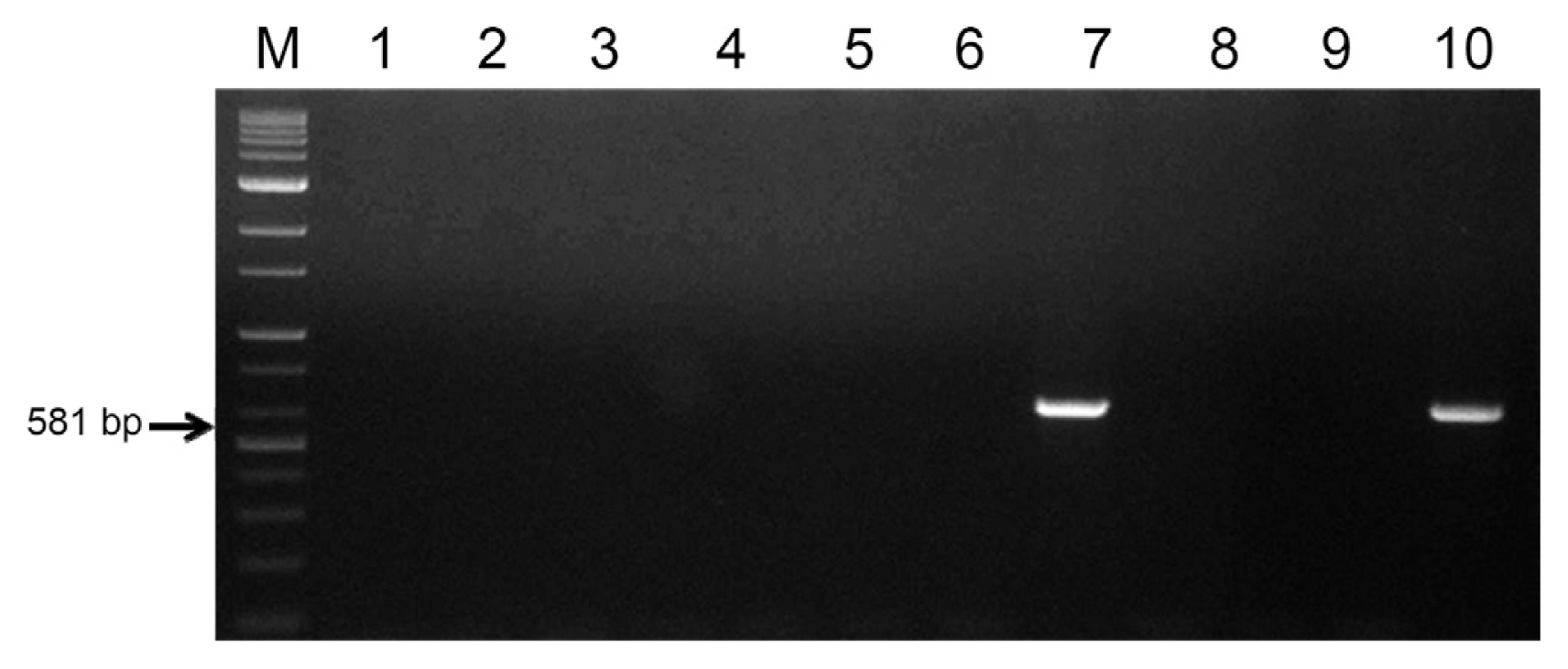
Table 1
Suppression of Xcc population in the suspension by irradiation with different X-ray doses
| Dose of X-ray (Gy) | Number of Xcca (Log cfu/ml) | Inhibition ratec (%) | Duncan’s testd (p=0.001) |
|---|---|---|---|
| control | 6.9 ± 0.2b | - | a |
| 50 | 5.5 ± 0.5 | 20.3 | b |
| 100 | 4.4 ± 0.5 | 36.2 | bc |
| 150 | 3.8 ± 0.3 | 44.9 | cd |
| 200 | 2.8 ± 0.6 | 59.1 | de |
| 250 | 2.0 ± 0.7 | 71.0 | e |
| 300 | 0.7 ± 0.5 | 89.9 | f |
| 350 | 0.4 ± 0.4 | 94.2 | f |
| 400 | 0.0 ± 0.0 | 100.0 | f |
Table 2
Suppression of Xcc population in the washing solution from Satsuma mandarin fruits with different doses of X-irradiation
| Dose of X-ray (Gy) | Number of Xcca (Log cfu/ml) | Inhibition ratec (%) | Duncan’s testd (p=0.001) |
|---|---|---|---|
| control | 3.1 ± 0.1b | - | a |
| 30 | 2.4 ± 0.3 | 22.6 | ab |
| 50 | 2.2 ± 0.2 | 29.0 | b |
| 100 | 1.6 ± 0.1 | 48.4 | c |
| 150 | 0.8 ± 0.3 | 74.2 | d |
| 200 | 0.2 ± 0.3 | 93.6 | d |
| 250 | 0.0 ± 0.0 | 100.0 | d |
| 300 | 0.0 ± 0.0 | 100.0 | d |
References
Abu-Tarboush, HM, Al-Kahtani, HA, Atia, M, Abou-Arab, AA, Bajaber, AS and El-Mojaddidi, MA 1997. Sensory and microbial quality of chicken as affected by irradiation and postirradiation storage at 4.0°C. J Food Prot. 60:761-770.



Ahn, HJ, Kim, JH, Kim, JK, Kim, DH, Yook, HS and Byun, MW 2005. Combined effects of irradiation and modified atmosphere packaging on minimally processed Chinese cabbage (Brassica rapa L.). Food Chem. 89:589-597.

Aquino, KAS 2012. Sterilization by gamma irradiation. In: Gamma Radiation, eds. by A Feriz, 171-206. InTech Press, Croatia.
Aquino, S, Ferreira, F, Ribeiro, DHB, Corrêa, B, Greiner, R and Villavicencio, ALCH 2005. Evaluation of viability of Aspergillus flavus and aflatoxins degradation in irradiated samples of maize. Braz J Microbiol. 36:352-356.

Bell, CH, Price, N and Chakrabarti, B 1996. The methyl bromide issue. Wiley and Sons, Chichester, UK. 400.
Cao, M, Fu, Y, Guo, Y and Pan, J 2009. Chlamydomonas (chlorophyceae) colony PCR. Protoplasma. 235:107-110.



Coletta-Filho, HD, Takita, MA, Souza, AA, Neto, JR, Destefano, SAL, Hartung, JS and Machado, MA 2006. Primers based on the rpf gene region provide improved detection of Xanthomonas axonopodis pv. citri in naturally and artificially infected citrus plants. J Appl Microbiol. 100:279-285.


Clavero, MR, Monk, JD, Beuchat, LR, Doyle, MP and Brackett, RE 1994. Inactivation of Escherichia coli O157: H7, Salmonellae, and Campylobacter jejuni in raw ground beef by gamma irradiation. Appl Environ Microbiol. 60:2069-2075.




Del Campo, R, Russi, P, Mara, P, Mara, H, Peyrou, M, De León, IP and Gaggero, C 2009. Xanthomonas axonopodis pv. citri enters the VBNC state after copper treatment and retains its virulence. FEMS Microbiol Lett. 298:143-148.


Dezordi, C, Maringoni, AC, Menten, JOM and Camara, RC 2009. Semi-selective culture medium for Xanthomonas axonopodis pv. malvacearum detection in cotton seeds (Gossypium hirsutum L.). Asian J Plant Pathol. 3:39-49.

Drake, S and Neven, L 1997. Quality response of ‘Bing’ and ‘Rainier’ sweet cherries to low dose electron beam irradiation. J Food Process Preserv. 21:345-351.

Elgazzar, AH and Kazem, N 2015. Biological effects of ionizing radiation. In: The pathophysiologic basis of nuclear medicine, eds. by AH Elgazzar, 369-370. Springer-Verlag, Berlin, Germany.

Fields, PG and White, NDG 2002. Alternatives to methyl bromide treatments for stored-product and quarantine insects. Annu Rev Entomol. 47:331-359.


Gareau, BJ 2010. A critical review of the successful CFC phase-out versus the delayed methyl bromide phase-out in the montreal protocol. Int Environ Agreements Polit Law Econ. 10:209-231.


Golmohammadi, M, Cubero, J, Peñalver, J, Quesada, J, López, M and Llop, P 2007. Diagnosis of Xanthomonas axonopodis pv. citri, causal agent of citrus canker, in commercial fruits by isolation and PCR-based methods. J Appl Microbiol. 103:2309-2315.


Gottwald, TR, Graham, JH and Schubert, TS 2002. Citrus canker: The pathogen and its impact. Plant Health Prog. 10:1-34.

Gottwald, TR, Hughes, G, Graham, JH, Sun, X and Riley, T 2001. The citrus canker epidemic in Florida: the scientific basis of regulatory eradication policy for an invasive species. Phytopathology. 91:30-34.


Graham, JH, Gottwald, TR, Cubero, J and Achor, DS 2004. Xanthomonas axonopodis pv. citri: Factors affecting successful eradication of citrus canker. Mol Plant Pathol. 5:1-15.



Grieb, TA, Forng, RY, Stafford, RE, Lin, J, Almeida, J, Bogdansky, S, Ronholdt, C, Drohan, WN and Burgess, WH 2005. Effective use of optimized, high-dose (50 kGy) gamma irradiation for pathogen inactivation of human bone allografts. Biomaterial. 26:2033-2042.

Hallman, GJ 2011. Phytosanitary applications of irradiation. Compr Rev Food Sci Food Saf. 10:143-151.

Han, SH, Jeun, YC and Song, SJ 2014. Changes of physical character and quality in Satsuma mandarin fruit irradiated by γ- and X-ray during low temperature storage. In: Physiology and Production, The third International Symposium on Citrus Biotechnology; Shizuoka. International Society For Horticultural Science (ISHS), Leuven, Belgium.
Hartung, JS, Daniel, JF and Pruvost, OP 1993. Detection of Xanthomonas campestris pv. citri by the polymerase chain reaction method. Appl Environ Microbiol. 59:1143-1148.




Jeong, JY, Yoon, MC, Lee, KY, Jung, K, Kim, HJ, Park, HJ and Jeong, RD 2014. Effect of the combined treatment with gamma irradiation and sodium dichloroisocyanurate on postharvest Rhizopus soft rot of sweet potato. J Plant Dis Prot. 121:243-249.


Jeong, RD 2014. Use of ionizing radiation as a phytosanitary treatment for postharvest disease control. J Radiat Ind. 8:97-104.
Kader, AA 1986. Potential applications of ionizing radiation in postharvest handling of fresh fruits and vegetables. Food Technol. 40:117-121.
Kim, JH and Yun, SC 2014 Effect of gamma irradiation and its convergent treatments on lily leaf blight pathogen, Botrytis elliptica, and the disease development. Res Plant Dis. 20:71-78 (in Korean).

Kim, KN, Song, MA, Han, SH, Song, SJ and Jeun, YC 2014 Inactivation of Xanthomonas citri subsp. citri and effect on infection of citrus canker by gamma irradiation. Res Plant Dis. 20:283-288 (in Korean).

Kositcharoenkul, N, Chatchawankanphanich, O, Bhunchoth, A and Kositratana, W 2011. Detection of Xanthomonas citri subsp. citri from field samples using single-tube nested PCR. Plant Pathol. 60:436-442.

Lee, NY, Jo, C, Shin, DH, Kim, WG and Byun, MW 2006. Effect of γ-irradiation on pathogens inoculated into ready-to-use vegetables. Food Microbiol. 23:649-656.


Mahmoud, GA, El-Tobgy, KM and Abo-El-Seoud, M 2010. Application of combined biocides and gamma radiation for keeping good quality stored grapefruits. Arch Phytopathol Plant Prot. 43:712-721.

Maity, JP, Kar, S, Banerjee, S, Sudershan, M, Chakraborty, A and Santra, SC 2011. Effects of gamma radiation on fungi infected rice (in vitro). Int J Radiat Biol. 87:1097-1102.


Mayer-Miebach, E, Stahl, M, Eschrig, U, Deniaud, L, Ehlermann, D and Schuchmann, H 2005. Inactivation of a non-pathogenic strain of E. coli by ionising radiation. Food Control. 16:701-705.

Mohammadi, M, Mirzaee, M and Rahimian, H 2001. Physiological and biochemical characteristics of iranian strains of Xanthomonas axonopodis pv. citri, the causal agent of citrus bacterial canker disease. J Phytopathol. 149:65-75.

Mostafavi, HA, Mirmajlessi, SM, Fathollahi, H, Minassyan, V and Mirjalili, SM 2013. Evaluation of gamma irradiation effect and pseudomonas flourescens against penicillium expansum. Afr J Biotechnol. 10:11290-11293.

Niemira, BA 2003. Irradiation of fresh and minimally processed fruits, vegetables and juices. In: The microbial safety of minimally processed foods, eds. by SN John, MS Gerald and KJ Vijay, 279-300. CRC Press, Florida, USA.

Osteen, C 2003. Methyl bromide phaseout proceeds: Users request exemptions. Amber Waves. 1:23-27.
Penner, JE 1999. Aviation and the global atmosphere. Cambridge University Press, UK. 333.
Schaad, NW, Postnikova, E, Lacy, G, Sechler, A, Agarkova, IV, Stromberg, PE, Stromberg, VK and Vidaver, AK 2006. Emended classification of xanthomonad pathogens on citrus. Syst Appl Microbiol. 29:690-695.


Schubert, TS, Rizvi, SA, Sun, X, Gottwald, TR, Graham, JH and Dixon, W (2001):Meeting the challenge of eradicating citrus canker in Florida-Again. Plant Dis. 85:340-356.


Sheu, DS, Wang, YT and Lee, CY 2000. Rapid detection of polyhydroxyalkanoate-accumulating bacteria isolated from the environment by colony PCR. Microbiology. 146:2019-2025.


Sommers, CH and Boyd, G 2006. Variations in the radiation sensitivity of food borne pathogens associated with complex ready-to-eat food products. Radiat Phys Chem. 75:773-778.

Stall, RE and Civerolo, EL 1991. Research relating to the recent outbreak of citrus canker in florida*. Annu Rev Phytopathol. 29:399-420.


Van Kooij, J, Leveling, H and Schubert, J 1978. Food Preservation by Irradiation. II: IAEA, Vienna, Austria. 63.
Vauterin, L, Hoste, B, Kersters, K and Swings, J 1995. Reclassification of Xanthomonas. Int J Syst Bacteriol. 45:472-489.

Watson, R, Albritton, D, Anderson, S and Lee-Bapty, S 1992. Methyl bromide: Its atmospheric science, technology, and economics. Montreal Protocol Assessment Suplement, United Nations Environment Programme. Nairobi, Kenya. 234.
World Health Organization (WHO). 1981. High dose irradiation. Wholesomeness of irradiated food. WHO Technical Report Series 659. Geneva, Switzerland.
Yoon, MC, Jung, K, Lee, KY, Jeong, JY, Lee, JW and Park, HJ 2014. Synergistic effect of the combined treatment with gamma irradiation and sodium dichloroisocyanurate to control gray mold (Botrytis cinerea) on paprika. Radiat Phys Chem. 98:103-108.

Youssef, B 1994. Microbial flora of frozen beef burger as affected by gamma radiation. Egypt J Microbiol. 29:105-113.
- TOOLS
-
METRICS

- Related articles
-
Investigation of Genetic Diversity of
Fusarium oxysporum f. sp.fragariae Using PCR-RFLP2017 ;33(2)



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



