 |
 |
| Plant Pathol J > Volume 38(4); 2022 > Article |
|
Abstract
Erwinia amylovora and E. pyrifoliae are the causative agents of destructive diseases in both apple and pear trees viz. fire blight and black shoot blight, respectively. Since the introduction of fire blight in Korea in 2015, the occurrence of both pathogens has been independently reported. The co-incidence of these diseases is highly probable given the co-existence of their pathogenic bacteria in the same trees or orchards in a city/district. Hence, this study evaluated whether both diseases occurred in neighboring orchards and whether they occurred together in a single orchard. The competition and virulence of the two pathogens was compared using growth rates in vitro and in planta. Importantly, E amylovora showed significantly higher colony numbers than E. pyrifoliae when they were co-cultured in liquid media and co-inoculated into immature apple fruits and seedlings. In a comparison of the usage of major carbon sources, which are abundant in immature apple fruits and seedlings, E. amylovora also showed better growth rates than E. pyrifoliae. In virulence assays, including motility and a hypersensitive response (HR), E. amylovora demonstrated a larger diameter of travel from the inoculation site than E. pyrifoliae in both swarming and swimming motilities. E. amylovora elicited a HR in tobacco leaves when diluted from 1:1 to 1:16 but E. pyrifoliae does not elicit a HR when diluted at 1:16. Therefore, E. amylovora was concluded to have a greater competitive fitness than E. pyrifoliae.
In Korea, Erwinia amylovora, the causal agent of fire blight in the family Rosaceae, was first reported in apple and pear trees in 2015 (Myung et al., 2016; Park et al., 2016). Subsequently, fire blight was detected between 2015 and 2017 in 43, 17, and 33 orchards covering 42.9, 15.1, and 22.7 ha, respectively. In 2018, the incidence increased to 67 orchards over 48.2 ha and then exponentially increased to 188, 744, and 618 orchards covering 131.5, 394.4, and 294.3 ha, respectively, between 2019 and 2021. The government attempted to intensively control fire blight since its first incidence by eradication so as to prevent its dispersal to new apple and pear orchards (Park et al., 2017) but this control strategy obviously was not effective.
E. pyrifoliae is a domestic pathogen which is the causative agent of black shoot blight in Asian pears in Korea (Kim et al., 1999; Shrestha et al., 2003). It was also reported to infect some apple cultivars by means of artificial inoculation in an experimental orchard (Kim et al., 2001). The symptoms of black shoot blight on leaves and shoots are very similar to those of fire blight, where necrotic strips on the veins of leaves spread to shoots resulting in necrosis of subsequent tissues, such as flowers and immature fruits. Because of the similar symptoms and morphological and biochemical characteristics of fire and black shoot blight in the early stages, several governors and researchers misconstrued the detection of fire blight in Korea at that time. However, experiments including phylogenetic analyses using 16S-23S intergenic transcribed spacer regions, plasmid profiles, and detection using species-specific primer sets have revealed that the black shoot blight and fire blight pathogens are different (Jock and Geider, 2004; Maxson-Stein et al., 2003; McGhee et al., 2002; Shrestha et al., 2007). The black shoot blight pathogen was finally identified as a novel species, E. pyrifoliae, based on name of its host, Pyrus pyrifolia (Kim et al., 1999; Rhim et al., 1999). Although Kim et al. (2001) and Lehman et al. (2008) reported that black shoot blight and its pathogen, E. pyrifoliae, were extensively found between 1995 and 1998 and were not produced or detected from 1999 to the 2000s, black shoot blight was present during that period.
E. amylovora originated from North America (Bonn and van der Zwet, 2000); E. pyrifoliae is native to Korea (Kim et al., 1999) and has not been detected elsewhere, except in strawberries in the Netherlands (Marcel and Maria, 2015). Fire blight and black shoot blight can therefore not be produced simultaneously in apple and pear trees in same region, indicating that two pathogens possibly cannot co-exist in nature. However, since the introduction of fire blight to Korea in 2015, both fire blight and black shoot blight have been detected in apple and pear orchards in Korea. This has occurred in exceptional cases in five cities/districts in three provinces and 13 cities/districts in five provinces in 2020 and 2021 where both diseases have been detected at least once in different orchards in same year or before/after a disease event. Recently, this has become more frequent. The original premise that both diseases are not produced in same area is likely inaccurate. This raises the possibility that both diseases will inevitably be detected on one tree of apples or pears in one orchard in Korea, which portends enormous industrial and economical losses. Therefore, the competitive fitness of the two pathogens needs to be evaluated. Thus, in this study, the characteristics of E. amylovora and E. pyrifoliae were compared in terms of growth rate, motility, carbon utilization, hypersensitive response (HR), and pathogenicity in immature apple fruits and seedlings.
Wild type E. amylovora TS3128 (Kang et al., 2021) and E. pyrifoliae Ep1 (Kim et al., 1999) used in this study were isolated from infected pears from Anseong and Chuncheon, Korea, in 2015 and 1996, respectively. For the experiments investigating the competitive fitness between the strains, a plasmid, pBAV1K-T5-gfp (Bryksin and Matsumura, 2010) or pW208, which harbored a kanamycin (50 μg/ml) or tetracycline (10 μg/ml) resistant gene, was transformed into TS3128 or Ep1, respectively, generating Ea-KmR or Ep-TcR.
To compare growth between wild type strains (E. amylovora and E. pyrifoliae) and the antibiotic-resistant strains, both strains were inoculated into 30 ml of mannitol glutamate yeast extract (MGY) broth with or without the corresponding antibiotics at an optical density (O.D.) at 600nm of 0.001 (1.2 × 105 colony forming units [cfu]/ml) and then incubated at 28°C for 48 h. The O.D.600nm values were measured at 12 h intervals. For analysis of the co-culture growth rates of the two pathogens, 15 ml of MGY broth that was inoculated at O.D.600nm = 0.001 with each strain was mixed together in 30 ml and incubated at 28°C for 48 h. An amount of 100 ml that was diluted 102 to 105-fold was plated on MGY medium supplemented with the corresponding antibiotic at 12 h intervals. Cfu/ml were counted after incubation at 28°C for 24 h. The experiment was performed twice.
Two kinds of plant material were used for competition between E. amylovora and E. pyrifoliae. Immature apple fruit (Malus domestica cv. Fuji) were sterilized with 70% ethanol and punctured using a no. 1 cork bore (diameter 0.5 cm, depth 1 cm). An amount of 100 μl of the bacterial suspension (O.D.600nm = 0.001) of each antibiotic-resistant strain or a 100 μl mixture of a 1:1 ratio of the two pathogens was inoculated into the hole and incubated at 28°C for 15 days. At 0, 1, 2, 4, 6, 10, 13, and 15 days post inoculation (dpi), a 1 cm2 portion was taken using a no. 4 cork bore and macerated in 10 mM MgCl2 buffer. An amount of 100 μl of the suspension was plated on MGY medium supplemented with kanamycin or tetracycline for co-incubation with Ea-KmR or Ep-TcR, respectively, and incubated at 28°C for 2 days, and the bacteria that grew on the plates were counted as cfu/ml.
Four-week-old seedlings of two apple rootstocks (M9 and M26) were inoculated by dipping in the individual or mixed bacterial suspension (O.D.600nm = 0.001) for 30 s and were incubated at 28°C for 10 days. Bacterial growth was measured at 0 and 10 dpi by grinding the whole seedlings in 10 mM MgCl2 buffer, and plating 10-fold dilutions in duplicate on MGY medium supplemented with the corresponding antibiotics. Plated MGY media were incubated at 28°C for 2 days and colonies were counted as cfu/ml. In planta experiments were conducted twice with two immature apple fruits and seedlings. The experiment was performed twice.
Metabolic activities of E. amylovora TS3128 and E. pyrifoliae Ep1 on 95 substrates were compared using a BIOLOG GN2 MicroPlate (BIOLOG Inc., Hayward, CA, USA) following the manufacturer’s protocol. Activities elicited by bacteria on substrates were monitored by naked eye, with color changes to violet, pale violet, or no color change indicating positive, variable, or negative reactions, respectively.
To analyze the major sugars in immature apple fruits and seedlings, immature fruits and seedlings were mixed with 80% methanol (1:1 ratio) and water (1:75 ratio), respectively, and mixtures were ground using a mixer (HMF-3260S, Hanil Elec. Co., Seoul, Korea). Ground mixtures were centrifuged at 4,000 rpm for 10 min and supernatants were filtered using Whatman ashless filter paper (90 mm diameter; Merck KGaA, Darmstadt, Germany). Filtrates were evaporated using a nitrogen concentrator and diluted 10 times with water and were purified with a Sep-Pak C18 cartridge. High performance liquid chromatography (Waters, Milford, MA, USA) was conducted using a Sugar Pak I column (10 mm, 6.5 mm × 300 mm) with 200 ml injection volume and water as eluent at a flow rate of 0.5 ml/min, with temperature of the column and detector at 90°C and 35°C, respectively. Contents of sucrose, glucose, and fructose were calculated using the standard curve to obtain areas under the curve.
The growth rates of E. amylovora TS3128 and E. pyrifoliae Ep1 were tested using three major carbon sources that are abundant in immature apple fruits and seedlings. Bacterial suspensions (O.D.600nm = 0.1, 3.1 × 108 cfu/ml) of the two pathogens were individually inoculated into liquid minimal medium (1 g NH4H2PO4, 0.2 g KCl, 0.2 g MgSO4·7H2O, 1 g/l peptone) supplemented with 0.1%, 0.5%, 1.0%, 1.5%, and 2.0% each of sucrose, glucose, and fructose at a final concentration of O.D.600nm = 0.005 (4 × 105 cfu/ml) and incubated at 28°C for 72 h. Growth rates were calculated using O.D.600nm at 12 intervals measured with a spectrophotometer (DU730, Beckman Coulter Inc., Krefeld, Germany). All experiments were performed twice.
Motilities were tested using swimming and swarming assays. Each bacterial suspension was prepared in 10 mM MgCl2 buffer at a concentration (O.D.600nm = 0.1) from grown overnight in MGY medium. A 5 μl suspension was spot-inoculated onto the left and right sides of a 0.3% agarose medium for swimming (Berry et al., 2009) or an agar plate for swarming (Wang et al., 2010) as described previously, and also spot-inoculated symmetrically on the left and right using TS3128 and Ep1, respectively. The diameters traveled from the inoculation site were measured using analog calipers after incubation at 28°C for 3 days. The experiment was performed twice.
Five-week-old tobacco plants (Nicotiana tabacum cv. Samsun) in a growth chamber were used for infiltration of each bacterial suspension (diluted to 1:1, 1:2, 1:4, 1:8, and 1:16 of O.D.600nm = 0.1), which was prepared in 10 mM MgCl2 buffer from grown overnight in MGY medium. Infiltrations were conducted into the third to sixth oldest leaves using a blunt syringe with these diluted bacterial suspensions. Inoculated plants were maintained at 28°C for 12 h then monitored at 2-h intervals for water-soak and necrosis to indicate HR development and photography.
Two replicates of the experiments were conducted for the determination of growth rate in liquid media and plant materials, usage of carbon sources, and motility tests. Data were analyzed using analysis of variance (ANOVA), and the means were compared using Duncan’s least significant difference test at significance levels of P < 0.01. The analysis was performed using SAS software (ver. 9.2, SAS Institute Inc., Cary, NC, USA).
Since the first report of fire blight in Korea, Korean governmental organizations, the Rural Development Administration (RDA), Animal and Plant Quarantine Agency (QIA), and plant pathologists at the University, have regularly monitored fire blight and black shoot blight four times a year nationwide, including orchards in which it had and had not been detected in the previous year. Fire blight was detected in only two cities (Anseong and Cheonan) from 2016 to 2017 but it had spread to six in 2018, and then 10, 15, and 22 cities/districts in 2019, 2020, and 2021, respectively (Table 1). Black shoot blight was detected in four cities in 2016, five in 2017, one in 2018, three in 2019, 17 in 2020, and six cities/districts in 2021 (Table 1). Importantly, both fire blight and black shoot blight occurred in 2020 in apple orchards in Yeoncheon, Anseong, Pyeongchang, Chungju, and Eumseong in 2020; and in apple or pear trees Wonju and Yeongju in 2021 (Table 1). The pattern of detection of fire blight from 2016 to 2021 indicates that it most likely spread from the original cities/districts in 2015 to the neighboring cities/districts (Fig. 1). Black shoot blight was limited to the northern parts of Gyeonggi-do and Gangwon-do, except for recent detection in Chungju, Yeongju, and Mungyeong (Fig. 1).
The kanamycin or tetracycline-resistant strains exhibited similar growth rates in comparison to the wild type TS3128 or Ep1 at 28°C for 48 h in MGY broth culture (Fig. 2A). Therefore, each antibiotic-resistant strain was appropriated for use in the experiments of competitive fitness. However, when they were co-cultured, Ep-TcR was shown to reduce cfu (P < 0.005) compared to Ea-KmR, whereas individual culture of each strain was had similar growth (Fig. 2B).
In immature apple fruits, Ea-KmR demonstrated higher growth than Ep-TcR when individually inoculated. The cfus between Ea-KmR and Ep-TcR differed greatly at the observed times when co-inoculated into one fruit (Fig. 3A). In addition, Ea-KmR and Ep-TcR had similar colony numbers with individual inoculation of both varieties of apple seedlings. Colonies of Ep-TcR were lower than those of Ea-KmR when they were co-inoculated into seedlings regardless of the variety of apple (Fig. 3B and C). The symptoms were developed 4-5 days post inoculation both immature fruits and seedlings. These patterns were consistent in immature apple fruits and seedlings.
Of the 95 substrates in BIOLOG GN2 MicroPlate, utilization of most carbon and nitrogen sources were exactly the same by both TS3128 and Ep1. Several differences in metabolic activity in carbon sources including cellobiose, gentiobiose, and salicin, and nitrogen sources such as histidine, serine, and sodium butyrate were evident (Table 2).
Generally, the most abundant sugars in plant materials such as immature apple fruits and seedlings are sucrose, glucose, and fructose. These sugars are also known to be the most important resources for bacterial growth. Fructose was the most plentiful sugar in immature apple fruits, whereas glucose was the highest in apple seedlings, regardless of the apple varieties tested in this study (Table 3). Interestingly, TS3128 had greater ability than Ep1 to use all three sugars. Utilization was not shown to be different using the BIOLOG GN2 MicroPlate (Fig. 4), indicating that the sugars were used as a sole carbon source with greater ability by E. amylovora than E. pyrifoliae.
In motility tests, TS3128 traveled a longer distance from the inoculation site than Ep1 in both swimming and swarming assays. Both pathogens were able to swim further than swarm. Based on the similar diameter of TS3128 or Ep1 inoculated simultaneously on the left and right sides, no antagonisms were observed between TS3128 and Ep1 (Fig. 5).
A difference or a strongly impaired ability to elicit HR in tobacco leaves between TS3128 and Ep1 was not observed. In repeated assays, however, the 1:16 dilution of the original suspension of Ep1 impaired the HR relative to TS3128 (Fig. 6). No difference 1:2, 1:4, and 1:8 bacterial dilutions of TS3128 and Ep1 in five replications in the older, lower leaves and younger, upper leaves.
Dispersal of fire blight likely originated from the first reported sites, such as Anseong, Cheonan, and Jecheon, to cities/districts in their vicinity, indicating that fire blight is spread via biotic factors such as insects and human activities rather than abiotic factors. These possibilities were explored in previous reports; E. amylovora can survive on the surface of clothes, pruning shears, and rubber boots for several days and up to 24 h, respectively (Choi et al., 2019) and it can survive for longer periods than previously found on the surface and interior of honeybees (Choi et al., 2022). Therefore, biotic factors may have role importantly in central regions of Korea for dissemination of fire blight. In contrast, the occurrence of black shoot blight can be restricted to the northern parts of Gyeonggi-do and Gangwon-do. Lower than average temperatures in this region than in the southern parts and other provinces is considered the main reason, because E. pyrifoliae is more cold-tolerant than E. amylovora, based on the higher growth rates of E. pyrifoliae than those of E. amylovora at 12-21°C (Shrestha et al., 2005). However, production of black shoot blight occurred towards the south where average temperatures are higher than the original sites, suggesting that E. pyrifoliae may adapt to high temperatures or frequent human activities such as hand-pruning and thinning may contribute to its spread into new cities/districts. Thus, co-infection with both diseases is likely to occur in the near future. As shown in Table 1 and Fig. 1, co-incidence of fire blight and black shoot blight has been detected from 2020 to the present.
The distance of the closest orchards in which fire blight and black shoot blight were detected 5.3 km in Pyeongchang (37°21′23.5″N, 128°24′21.4″E and 37°23′33.3″N, 128°21′55.9″E), 13.4 km in Yeoncheon (38°1′27.5″N, 126°59′36.2″E and 38°7′16.4″N, 127°5′12.4″E), and in two adjacent orchards in Chungju (37°8′50.6″N, 127°42′20.7″E and 37°8′56.5″N, 127°42′26.1″E), with a different subdivision in Eumseong. In 2021, the closest distance between fire blight and black shoot blight was 0.08 km and 1.2 km in Wonju (37°9′41.7″N, 127°51′7.5″E and 37°9′47.7″N, 127°51′1.5″E) and Yeongju (37°0′28.9″N, 128°40′16.8″E and 37°0′35.3″N, 128°39′26.2″E), respectively. However, both diseases were detected in one orchard (36°57′59.3″N, 127°16′33.3″E) in Anseong, 2020, but fortunately, the infected pear trees had different disease. Currently, therefore, orchards with the simultaneous infection of both diseases seem to be closer than was the case in the 2010s.
The high possibility of the co-existence or co-incidence of both pathogens in Korea necessitates the determination of physiologic and pathogenic properties. Since the 2008 study of Lehman et al. on the interaction between E. amylovora and E. pyrifoliae, no more studies on site differentiation for either pathogen have been conducted. In Lehman et al. (2008), differentiation between E. amylovora and E. pyrifoliae was based on smaller population sizes deduced by the relative growth performance index of E. pyrifoliae and E. amylovora co-infected on Bartlett pear blossoms. They optimized the quantitative analysis of the pathogens using real-time polymerase chain reaction, and based the difference on Ct values and standard curves constructed with serially diluted bacterial suspensions. Thus, in the current study, competition of pathogens was compared directly by co-infection in liquid culture and expanded to plant materials such as immature apple fruits and seedlings. Clearly, E. amylovora showed better competitive fitness than E. pyrifoliae when they were co-cultured in liquid culture (Fig. 2B) and in planta immature fruits and seedlings (Fig. 3). Both TS3128 and Ep1 strains have been isolated from pear trees (Kang et al., 2021; Kim et al., 1999), but plant materials such as immature apple fruits and seedlings were used in planta in this study. Therefore, the different ability of the two pathogens to grow on plant materials could be due to the alternative host plant material; however, this hypothesis is rejected because two pathogens show similar growth on either plant material (Fig. 3).
Another explanation for the different competitive ability of the two pathogens can also be the metabolism of nutrient resources, including levan. However, there were no differences in the utilization of major carbon and nitrogen sources such as maltose, trehalose, sucrose, glucose, fructose, gluconate, pyruvate, and lactate based on the GN2 MicroPlate. In contrast, TS3128 showed higher growth rates than Ep1 in the usage of sucrose, glucose, and fructose as a sole carbon source, ranging from 0.1% to 2.0% in the liquid growth test (Fig. 4). The discrepancy in the utilization of major sugars between the BIOLOG GN2 MicroPlate and liquid growth may be a result of the method: the BIOLOG GN2 MicroPlate yields a metabolic fingerprint using tetrazolium violet as a redox dye to colorimetrically visualize the reaction. Thus, positive results shown by the GN2 MicroPlate may be induced by two-fold differences in O.D. values. This is important because the three kinds of major sugars were abundant in immature apple fruits and seedlings in the current study (Table 3). Similar abundance of sucrose is seen in blossoms (Bieleski, 1969). The population of Ep1 was smaller than TS3128 when inoculated together in immature fruits and seedlings (Fig. 3), which indicates that metabolic differences in the two pathogens with respect to carbon sources may be the reason for their different ability to colonize in/on host tissues.
Another factor seems to be amylovoran, which is a heteropolymer consisting of galactose, glucose, and pyruvate (Nimtz et al., 1996). This was determined to be a pathogenic factor according to the change to avirulence in amylovoran-deficient mutants (Steinberger and Beer, 1988). Although the amount of amylovoran produced by each pathogen was not quantified or compared, the higher utilization of sucrose, glucose, and fructose in TS3128 may be related to the production of more of this pathogenic factor in TS3128. Additionally, levan seems to be a factor in the competition between the two pathogens. Levan is a homopolymer of fructose and is synthesized following the breakdown of sucrose by levansucrase, encoded by the lsc gene (Gross et al., 1992). However, this gene is absent in E. pyrifoliae DSM 12163T (Smits et al., 2010a, 2010b), which was isolated with Ep1 from the same host and geographical site in 1996 but is present in TS3128. Therefore, TS3128 may be able to colonize in/on plant tissues using levansucrase, producing levan from sucrose that exist abundantly in immature apple fruits and seedlings.
Although the genes encoding flagellar biosynthesis and chemotaxis-related proteins in TS3128 and Ep1 were not analyzed in the current study, E. amylovora CFBP 1430 and E. pyrifoliae DSM 12163T have two set of genes encoding flagellar biosynthesis and chemotaxis-related proteins (Smits et al., 2010a, 2010b). Furthermore, both TS3128 and Ep1 have been routinely observed by several researchers to have peritrichous flagella. This suggests that E. amylovora TS3128 and E. pyrifoliae Ep1 in the current study should have genes encoding flagellar biosynthesis and chemotaxis-related proteins. Swarming and swimming are defined as multicellular movement on a surface and individual movement in liquid, respectively, but either motility is powered by rotating helical flagella (Kearns, 2010). The smaller diameters of swimming or swarming in Ep1 than TS3128 are inexplicable, but this does not eliminate the possibility that genes encoding related flagellar biosynthesis and chemotaxis may play a role in the different motilities of the two pathogens. A greater motility in TS3128 than Ep1 should therefore be correlated to stronger competition in E. amylovora than E. pyrifoliae (Fig. 5).
The elicitation of the HR depends on the hypersensitive reaction and pathogenicity (hrp) system during the early infection times of the interactions between microbes and plants (Alfano and Collmer, 2004). Similarity in the hrp region of E. pyrifoliae DSM 12163T and E. amylovora Ea321 has been reported (Oh et al., 2005), except for the absence of ORF1 and ORF2 in DSM 12163T (Smits et al., 2010a). However, both DSM 12163T and Ep1 had been reported to elicit HR in tobacco leaves except in some avirulent strains of E. pyrifoliae isolated from the thsame host (Jock et al., 2003). Thus, the impaired HR of the diluted Ep1 suspension may not evoke differential virulence because of the ability to induce different degrees of HR in E. amylovora strains (Wang et al., 2010). The reduced HR of Ep1 compared to TS3128 at a 1:16 dilution of the suspension (O.D600nm = 0.1) may be due to the absence of ORF1 and ORF2 or another unknown T3SS apparatus and effector protein. Ep1 at diluted concentrations shows a clearly reduced HR compared to TS3128 (Fig. 6) and this difference may be correlated to the greater competitive ability of E. amylovora over E. pyrifoliae.
In conclusion, the results indicate that E. amylovora TS3128 demonstrates greater competitive fitness than E. pyrifoliae Ep1 in terms of co-culture in liquid, co-inoculation of immature apple fruits and seedlings, growth rate using major carbon sources, motility, and HR. It is possible that fire blight and black shoot blight can be detected in the same region and their pathogens can exist simultaneously. This information must inform treatment and prevention protocol for both pathogens.
Acknowledgments
This research was supported by the Rural Development Administration (PJ015302012022), Republic of Korea.
Fig. 1
Distribution of fire blight and black shoot blight among cities and districts in Korea (2016-2021). Colored cities/districts imply accumulated production of diseases in each year. Red, gray, and yellow shading indicate cities/districts where fire blight, black shoot blight, or both diseases were reported, respectively.

Fig. 2
Effects of strain and co-culture on the growth rates of Erwinia amylovora TS3128 and E. pyrifoliae Ep1 in mannitol glutamate yeast extract broth. (A) Growth rates of separately cultured wild type strains (Ea-TS3128 and Ep-1/96) and antibiotic-resistant strains (Ea-KmR and Ep-TcR). (B) Growth rates of antibiotic-resistant strains when separately cultured (Ea-KmR and Ep-TcR) or co-cultured (C_Ea-KmR and C_Ep-TcR). Growth rates of Ea-KmR and Ep-TcR differed significantly at every time point (P < 0.005, Duncan’s least significant difference test). Values and error bars represent mean ± standard deviation (n = 2).
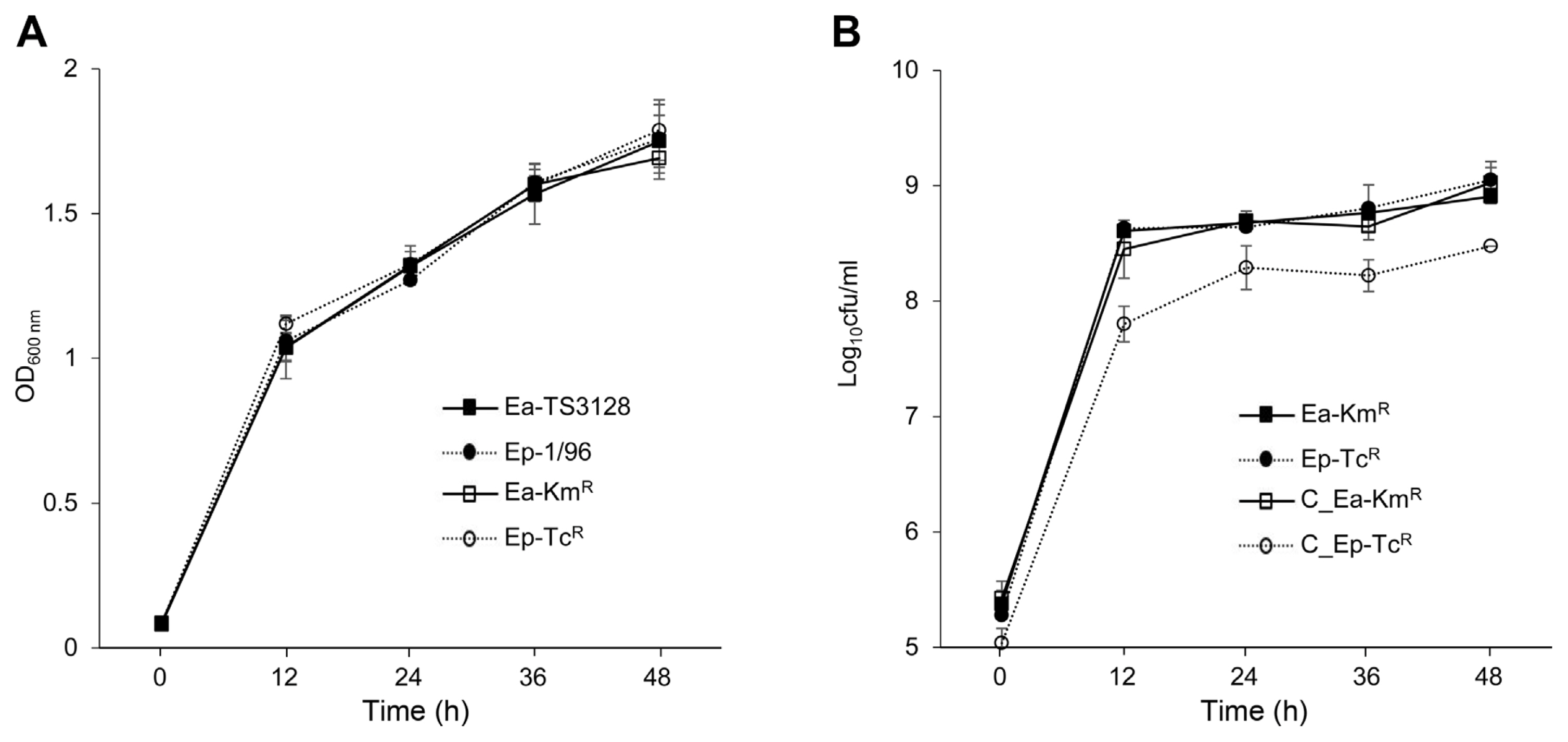
Fig. 3
In planta growth rates of antibiotic-resistant Erwinia amylovora TS3128 (Ea) and E. pyrifoliae Ep1 (Ep) in planta conditions. (A) Growth rates of antibiotic-resistant strains when separately inoculated (Ea-KmR and Ep-TcR) or co-inoculated (C_Ea-KmR and C_Ep-TcR) into immature apple fruits (Malus demestica cv. Fuji). (B, C) Density of antibiotic-resistant strains after 10 days when separately inoculated (Ea-KmR and Ep-TcR) or co-inoculated (C_Ea-KmR and C_Ep-TcR) onto apple seedlings (M9 and M26 rootstock). Density of co-inoculated Ep-TcR (C-Ep-TcR) were consistently lower than those of Ea-KmR (C_E-KmR) on both immature fruits (P < 0.005) and seedlings (P < 0.0001, Duncan’s least significant difference test). Values and error bars represent mean ± standard deviation (n = 2).
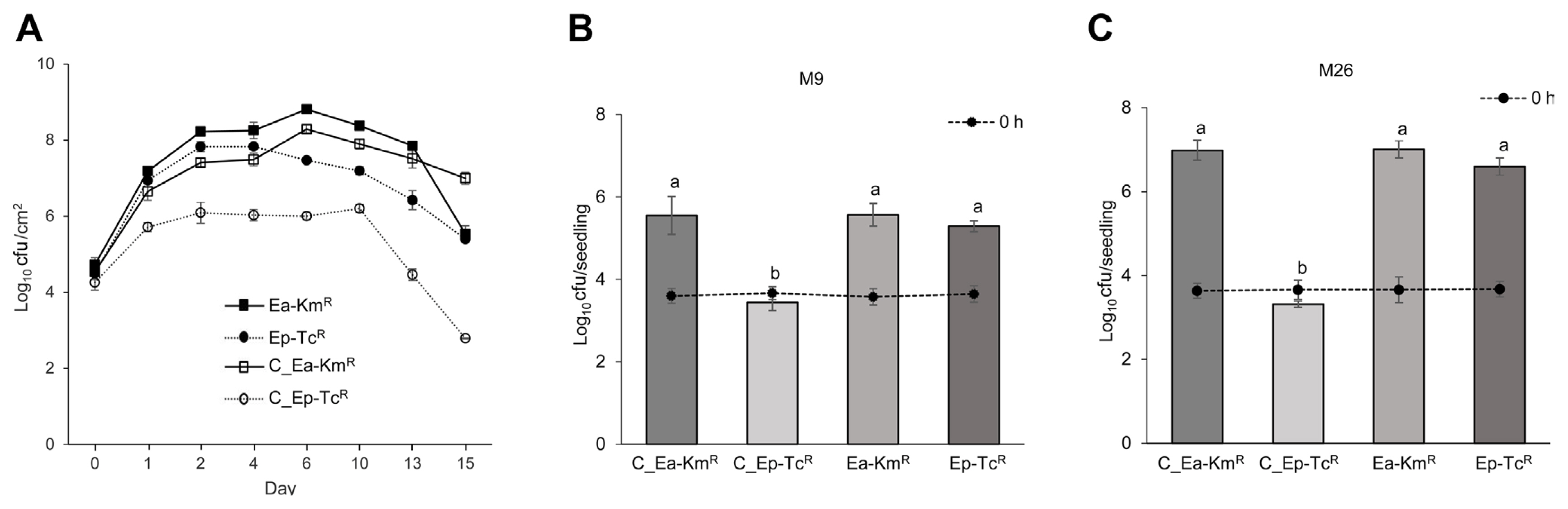
Fig. 4
Utilization of sole carbon sources in minimal liquid media by Erwinia amylovora TS3128 (Ea) and E. pyrifoliae Ep1 (Ep). Media was supplemented with each sugar (A, fructose; B, glucose; C, sucrose) at 0.1%, 0.5%, 1.0%, 1.5%, and 2%. The O.D. values of TS3128 and Ep1 differed significantly at 72 h post inoculation (P < 0.005, Duncan’s least significant difference test). Values and error bars represent mean ± standard deviation (n = 2).
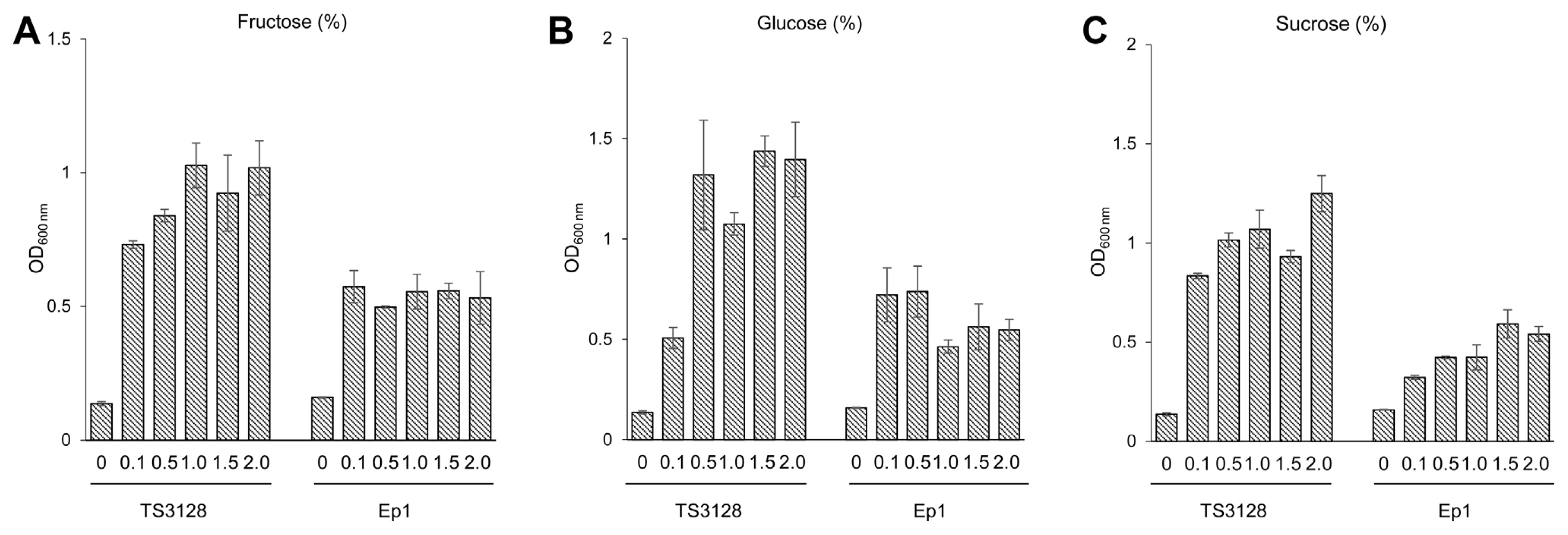
Fig. 5
Motilities of Erwinia amylovora TS3128 (Ea) and E. pyrifoliae Ep1 (Ep). (A) Swarming motility comparison. (B) Swimming motility comparison. Values and error bars indicate are the mean ± standard deviation (n = 2) distances from plates with single strains. The diameters of TS3128 and Ep1 differed significantly (P < 0.0001, Duncan’s least significant difference test). Center plates shown no antagonism between two pathogens.

Fig. 6
Hypersensitive response of tobacco plants (Nicotiana tabacum cv. Samsun) to diluted suspensions of Erwinia amylovora TS3128 and E. pyrifoliae Ep1. Images illustrate the response of tobacco leasves to 1:2, 1:4, 1:8, and 1:16 dilution of O.D.600 nm = 0.1 bacterial suspensions after 12 h infiltration.

Table 1
Cities/districts in Korea with orchards in which fire blight and black shoot blight were detected in apple or pear trees from 2016 to 2021
Table 2
Differences in metabolic activities in Erwinia amylovora TS3128 and E. pyrifoliae Ep1 in BIOLOG GN2 MicroPlate
| Resource | E. amylovora TS3128 | E. pyrifoliae Ep1 |
|---|---|---|
| D-Cellobiose | Δa | − |
| Gentiobiose | + | − |
| D-Salicin | Δ | − |
| L-Histidine | − | + |
| L-Serine | − | + |
| Sodium butyrate | Δ | − |
Table 3
Quantities of sucrose, glucose, and fructose in immature apple fruits (Malus demestica cv. Fuji) and seedlings (M9 and M26 rootstock) on a fresh-weight basis
| Sample | Type of sugar | Quantity (mg/g) |
|---|---|---|
| Immature fruit | Sucrose | 5.28 ± 0.19 |
| Glucose | 6.94 ± 0.46 | |
| Fructosea | 8.91 ± 0.67 | |
| Seedling | ||
| M9 | Sucrose | 8.32 ± 0.08 |
| Glucosea | 26.28 ± 1.14 | |
| Fructose | 5.10 ± 0.58 | |
| M26 | Sucrose | 5.84 ± 0.41 |
| Glucosea | 17.53 ± 0.38 | |
| Fructose | 6.44 ± 0.37 | |
References
Alfano, J.R. and Collmer, A. 2004. Type III secretion system effector proteins: double agents in bacterial disease and plant defense. Annu. Rev. Phytopathol 42:385-414.


Berry, M.C., McGhee, G.C., Zhao, Y. and Sundin, G.W. 2009. Effect of a waaL mutation on lipopolysaccharide composition, oxidative stress survivals, and virulence in Erwinia amylovora
. FEMS Microbiol. Lett 291:80-87.

Bieleski, R.L. 1969. Accumulation and translocation of sorbitol in apple phloem. Aust. J. Biol. Sci 22:611-620.

Bonn, W.G. and van der Zwet, T. 2000. Distribution and economic importance of fire blight. In: Fire blight: the disease and its causative agent, Erwinia amylovora
, eds. by J.L. Vanneste, pp. 37-53. CABI Publishing, Wallingford, UK.

Bryksin, A.V. and Matsumura, I. 2010. Rational design of a plasmid origin that replicates efficiently in both gram-positive and gram-negative bacteria. PLoS ONE 5:e13244.



Choi, H.J., Kim, Y.J., Lim, Y.-J. and Park, D.H. 2019. Survival of Erwinia amylovora on surfaces of materials used in orchards. Res. Plant Dis 25:89-93.


Choi, H.J., Kim, Y.J. and Park, D.H. 2022. Extended longevity of Erwinia amylovora vectored by honeybees under in vitro conditions and its capacity for dissemination. Plant Pathol 71:762-771.


Gross, M., Geier, G., Rudolph, K. and Geider, K. 1992. Levan and levansucrase synthesized by the fireblight pathogen Erwinia amylovora
. Physiol. Mol. Plant Pathol 40:371-381.

Jock, S. and Geider, K. 2004. Molecular differentiation of Erwinia amylovora strains from North America and of two Asian pear pathogens by analyses of PFGE patterns and hrpN genes. Environ. Mirobiol 6:480-490.

Jock, S., Kim, W.-S., Barny, M.-A. and Geider, K. 2003. Molecular characterization of natural Erwinia pyrifoliae strains deficient in hypersensitive response. Appl. Environ. Microbiol 69:679-682.




Kang, I.-J., Park, D.H., Lee, Y.-K., Han, S.-W., Kwak, Y.-S. and Oh, C.-S. 2021. Complete genome sequence of Erwinia amylovora strain TS3128, a Korean strain isolated in an Asian pear orchard in 2015. Microbiol. Resour. Announc 10:e00694-21.




Kim, W.-S., Gardan, L., Rhim, S.-L. and Geider, K. 1999.
Erwinia pyrifoliae sp. nov., a novel pathogen that affects Asian pear trees (Pyrus pyrifolia Nakai). Int. J. Syst. Bacteriol 49:899-905.


Kim, W.S., Jock, S., Paulin, J.-P., Rhim, S.-L. and Geider, K. 2001. Molecular detection and differentiation of Erwinia pyrifoliae and host range analysis of the Asian pear pathogen. Plant Dis 85:1183-1188.


Lehman, S.M., Kim, W.-S., Castle, A.J. and Svircev, A.M. 2008. Duplex real-time polymerase chain reaction reveals competition between Erwinia amylovora and E. pyrifoliae on pear blossoms. Phytopathology 98:673-679.


Marcel, W. and Maria, B.-V. 2015.
Erwinia pyrifoliae, a new pathogen on strawberry in the Netherlands. J. Berry Res. 5:17-22.
Maxon-Stein, K., McGhee, G.C., Smith, J.J., Jones, A.L. and Sundin, G.W. 2003. Genetic analysis of a pathogenic Erwinia spp. isolated from pear in Japan. Phytopathology 93:1393-1399.


McGhee, G.C., Schnabel, E.L., Maxson-Stein, K., Jones, B., Stromberg, V.K., Lacy, G.H. and Jones, A.L. 2002. Relatedness of chromosomal and plasmid DNAs of Erwinia pyrifoliae and Erwinia amylovora
. Appl. Environ. Microbiol 68:6182-6192.




Myung, I.-S., Lee, J.-Y., Yun, M.-J., Lee, Y.-H., Lee, Y.-K., Park, D.H. and Oh, C.-S. 2016. Fire blight of apple, caused by Erwinia amylovora, a new disease in Korea. Plant Dis. 100:1774.

Nimtz, M., Mort, A., Domke, T., Wray, V., Zhang, Y., Qiu, F., Coplin, D. and Geider, K. 1996. Structure of amylovoran, the capsular exopolysaccharide from the fire blight pathogen Erwinia amylovora
. Carbohydr. Res 287:59-76.


Oh, C.-S., Kim, J.F. and Beer, S.V. 2005. The Hrp pathogenicity island of Erwinia amylovora and identification of three novel genes required for systemic infection. Mol. Plant Pathol 6:125-138.


Park, D.H., Lee, Y.-G., Kim, J.-S., Cha, J.-S. and Oh, C.-S. 2017. Current status of fire blight caused by Erwinia amylovora and action its management in Korea. J. Plant Pathol 99:59-63.
Park, D.H., Yu, J.-G., Oh, E.-J., Han, K.-S., Yea, M.C., Lee, S.J., Myung, I.-S., Shim, H.S. and Oh, C.-S. 2016. First report of fire blight disease on Asian pear caused by Erwinia amylovora in Korea. Plant Dis. 100:1946.

Rhim, S.L., Volksch, B., Gardan, L., Paulin, J.P., Langlotz, C., Kim, W.-S. and Geider, K. 1999.
Erwinia pyrifoliae, an Erwinia species different from Erwinia amylovora, causes a necrotic disease of Asian pear trees. Plant Pathol 48:514-520.

Shrestha, R., Koo, J.H., Park, D.H., Hwang, I., Hur, J.H. and Lim, C.K. 2003.
Erwinia pyrifoliae, a causal endemic pathogen of shoot blight of Asian pear tree in Korea. Plant Pathol. J 19:294-300.

Shrestha, R., Lee, S.H., Hur, J.H. and Lim, C.K. 2005. The effects of temperature, pH, and bactericides on the growth of Erwinia pyrifoliae and Erwinia amylovora
. Plant Pathol. J 21:127-131.

Shrestha, R., Lee, S.H., Kim, J.E., Wilson, C., Choi, S.-G., Park, D.H., Wang, M.H., Hur, J.H. and Lim, C.K. 2007. Diversity and detection of Korean Erwinia pyrifoliae strains as determined by plasmid profiling, phylogenetic analysis and PCR. Plant Pathol 56:1023-1031.

Smits, T.H.M., Jaenicke, S., Rezzonico, F., Kamber, T., Goesmann, A., Frey, J.E. and Duffy, B. 2010a. Complete genome sequence of the fire blight pathogen Erwinia pyrifoliae DSM 12163T and comparative genomic insights into plant pathogenicity. BMC Genomics 11:2.




Smits, T.H.M., Rezzonico, F., Kamber, T., Blom, J., Goesmann, A., Frey, J.E. and Duffy, B. 2010b. Complete genome sequence of the fire blight pathogen Erwinia amylovora CFBP 1430 and comparison to other Erwinia spp. Mol. Plant-Microbe Interact 23:384-393.


- TOOLS
-
METRICS

- Related articles
-
Isolation of Nine Bacteriophages Shown Effective against Erwinia amylovora in Korea2022 June;38(3)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



