 |
 |
| Plant Pathol J > Volume 38(4); 2022 > Article |
|
Abstract
Apple stem grooving virus (ASGV) is a destructive viral pathogen of pome fruit trees that causes significant losses to fruit production worldwide. Obtaining ASGV-free propagation materials is essential to reduce economic losses, and accurate and sensitive detection methods to screen ASGV-free plantlets during in vitro propagation are urgently necessary. In this study, ASGV was sensitively and accurately quantified from in vitro propagated apple plantlets using a reverse transcription droplet digital polymerase chain reaction (RT-ddPCR) assay. The optimized RT-ddPCR assay was specific to other apple viruses, and was at least 10-times more sensitive than RT-real-time quantitative PCR assay. Furthermore, the optimized RT-ddPCR assay was validated for the detection and quantification of ASGV using micropropagated apple plantlet samples. This RT-ddPCR assay can be utilized for the accurate quantitative detection of ASGV infection in ASGV-free certification programs, and can thus contribute to the production of ASGV-free apple trees.
Apple (Malus domestica) belongs to the family Rosaceae and is a widely grown and economically important fruit worldwide. Apples are one of the most popular fruits in Korea, with a total cultivated area of 34,359 ha and production of 515,931 tons (Korean Statistical Information Service, 2021). To increase genetic diversity, breeding programs require multiple apple cultivars. However, apple trees in Korea are susceptible to infection by several viruses, such as apple stem grooving virus (ASGV), apple chlorotic leaf spot virus (ACLSV), apple stem pitting virus (ASPV), and apple mosaic virus (Lee et al., 2020). Despite being asymptomatic, these viruses significantly reduce fruit yield and quality, especially through mixed infections (Lee et al., 2020). Among them, ASGV has the highest infection rate in cultivated apple trees in Korea; it is distributed worldwide and has been reported in a wide range of hosts, including pear, apricot, cherry, lily, and soybean (Bhardwaj and Hallan, 2019; Lee et al., 2020).
Availability of virus-free propagation materials is important for increasing the productivity of apple fruits. However, viruses infecting apple trees are naturally transmitted by infected propagation materials, which transmit the virus from generation to generation (Hu et al., 2018). Thus, the use of certified, virus-free propagation materials is essential for producing healthy fruit trees.
Developing sensitive detection methods is vital for the successful screening of virus-free propagation materials for apple trees. To date, several molecular detection methods, such as reverse transcription-polymerase chain amplification (RT-PCR) (Nabi et al., 2022), RT-real-time quantitative PCR (RT-qPCR) (Malandraki et al., 2017), loop-mediated isothermal amplification (Lu et al., 2018), recombinase polymerase amplification (Jeong et al., 2021; Kim et al., 2019), and CRISPR-based assays (Jiao et al., 2021), have been developed to detect apple tree viruses. Currently, conventional RT-PCR and RT-qPCR have been used by KoreaŌĆÖs Ministry of Agriculture, Food and Rural Affairs (KMAFRA), to screen for apple viruses for in vitro micropropagation of apple plants. Although promising for the routine testing of apple viruses, these methods generally are not suitable for quantifying viral loads from virus-infected samples. In addition, although RT-qPCR can quantify viral concentrations, the viral loads are calculated from amplification rates relative to a standard curve, rather than via absolute quantification (Vasudevan et al., 2021).
Digital PCR (dPCR) can be used for absolute quantification of target nucleic acid sequences without the need for a standard curve based on many partitioned reactions. dPCR includes droplet (droplet digital PCR [ddPCR]) and nanofluidic chips (chip-based dPCR) and quantifies target sequences based on the number of positive and negative partitions using Poisson statistics after the reaction is terminated (Dube et al., 2008). dPCR has several advantages over RT-PCR and RT-qPCR, including absolute estimates of target sequences and enhanced sensitivity, even if the target sequences have low concentrations (Hindson et al., 2011). In this study, we detected and quantified ASGV coat protein (CP) sequences using an RT-ddPCR assay for micropropagated apple shoots and highlighted its potential as a screening method for the production of ASGV-free plantlets.
Micropropagated ŌĆśHongroŌĆÖ apple plantlets were provided by the National Institute of Horticultural & Herbal Science, Korea. Total RNA was extracted from apple plantlet leaves and RNA quality and concentration were determined following the methodology of Kim et al. (2019). The RNA samples were first tested for ASGV presence using RT-PCR with previously used diagnostic primers (Shim et al., 2006). First-strand cDNA was synthesized using 2 ╬╝g total RNA, 0.5 ╬╝g random primer, 10 mM dNTP, RNase inhibitor (TaKaRa, Shinga, Japan), M-MLV reverse transcriptase (Promega, Madison, WI, USA) in 20 ╬╝l volume. The synthesized cDNAs were used as templates for RT-PCR, RT-qPCR, and RT-ddPCR analyses. To test for ASGV infection in these samples, RT-PCR was performed following the methodology of Kim et al. (2019). The RT-PCR products (approximately 714 bp) corresponding to the CP gene were determined using 1.5% agarose gel electrophoresis (data not shown).
To design specific primers and a probe for the RT-ddPCR assay, the complete CP sequences of 15 ASGV isolates (MG682506, MG682507, MG682508, MG682509, MG682510, GQ330294, KR606324, JX885582, KR815877, LC086300, LN90143, KX668488, LC184610, KX988001) from different countries, including Korea, were obtained from the GenBank database. The conserved sequences of the ASGV isolates were then determined using BioEdit version 7.0.5.3 (Supplementary Fig. 1). The forward, reverse, and probe sequences for detecting ASGV in RT-qPCR and RT-ddPCR assays are listed in Table 1.
RT-ddPCR was performed using a QX200 Droplet Digital PCR system (Bio-Rad Laboratories, Hercules, CA, USA). The reaction mixture consisted of 10 ╬╝l of 2├Ś ddPCR Supermix for Probes (Bio-Rad Laboratories), 900 nM each of the forward and reverse primers, 250 nM probe (Table 1), 4.9 ╬╝l DEPC-water, and 1 ╬╝l template cDNA in 20 ╬╝l reaction volume. The RT-ddPCR amplifications were performed following the methodology of Lee et al. (2021). The copy number of each sample was determined by the QuantaSoft program (Bio-Rad Laboratories). The individual droplet reactions were measured using a QX200 Droplet Reader and the positive and negative fluorescent signals were differentiated based on a set threshold. In total, 15,000 reacted droplets were used as reliable data for analyses.
To establish the optimal operating conditions for the RT-ddPCR assay for ASGV detection, eight temperature gradients from 48 to 62┬░C were used, and the temperature that maximally separated positive and negative fluorescence amplitude was determined as the optimum temperature; accordingly, an annealing temperature of 54┬░C was chosen for subsequent analyses (Fig. 1).
To determine the specificity of the RT-ddPCR assay for ASGV detection, major apple viruses reported in Korea, such as ACLSV, ASPV, and ASSVd (apple scar skin viroid), were used and non-template control (NTC) was used as a negative control. Only RNA samples obtained from ASGV-infected plant material showed separated positive droplet clusters, whereas RNA samples obtained from plants infected with other viruses and NTC showed no positive droplets. These results demonstrated that there was no cross-reactivity among viruses and verified the high specificity of the RT-ddPCR assay for ASGV detection (Fig. 2).
The sensitivity of RT-ddPCR was evaluated against RT-qPCR in triplicate using 10-fold serial dilutions of ASGV transcripts, with concentrations ranging from 1.7 ├Ś 106 to 1.7 ├Ś 103. ASGV transcripts were produced using a previously described method (Kim et al., 2019). The concentration of transcripts, referred to as the copy number, was calculated using the following formula: transcript concentration (╬╝g/╬╝l)/fragment size (bp) ├Ś 182.5 ├Ś 1013 (Fronhoffs et al., 2002). RT-qPCR was performed using following cycling steps: denaturation at 95┬░C for 10 min and 40 cycles at 95┬░C for 15 s, 60┬░C for 60 s, and 72┬░C for 30 s in three steps (Lee et al., 2021). The copy number of ASGV transcripts from each sample was determined by interpolating individual Cq values from a standard curve and using the corresponding log values of the ASGV transcript copy number (Pfaffl and Hageleit, 2001). The RT-qPCR results determined the copy number of ASGV transcripts as 1.7 ├Ś 105 copies/╬╝l (28.10 copies/╬╝l), whereas RT-ddPCR detected 1.7 ├Ś 104 copies/╬╝l (3.73 copies/╬╝l), indicating that RT-ddPCR was approximately 10 times more sensitive than RT-qPCR. Furthermore, the coefficient of variation values derived from the RT-qPCR and RT-ddPCR data ranged from 1.52-2.24% and 7.01-22.62%, respectively (Table 2).
To further test RT-ddPCR assay performance for detecting and quantifying ASGV, 37 apple leaf samples from in vitro propagated apple plantlets were evaluated using RT-qPCR and RT-ddPCR assays. ASGV was detected in 32 (86.5%) and 34 (91.9%) of the 37 apple plantlet samples by RT-PCR and RT-qPCR, respectively (Table 3, Supplementary Fig. 2). In contrast, RT-ddPCR determined 35 (94.6%) samples to be positive for ASGV (Table 3). One sample (no. 16) tested negative for ASGV by RT-PCR and RT-qPCR, but positive by RT-ddPCR. These findings indicate that the RT-ddPCR assay is more accurate than conventional RT-PCR assays and would be more reliable for detecting ASGV-infected samples containing low viral titers. However, two samples (nos. 28 and 31) were identified as ASGV-free apple plantlets by RT-PCR, RT-qPCR, and RT-ddPCR (Fig. 3).
ASGV infects Rosaceae fruit trees, including apple, pear, apricot, and cherry, and is considered a high-risk viral pathogen during cultivation of fruit trees and production of ASGV-free plantlets by KMAFRA (Lee et al., 2020). Considering the increasing economic impact of ASGV in the global fruit industry, a range of RT-PCR and TaqMan probe-based RT-qPCR assays have been developed as reliable and sensitive detection methods for monitoring ASGV infections in fruit trees and for ASGV-free certification programs (Malandraki et al., 2017; Nabi et al., 2022).
In this study, the ddPCR technique was first used to detect and quantify ASGV in in vitro micropropagated apple shoots. Although ddPCR has some disadvantages compared to qPCR, such as the requirement of more expensive instrumentation and reagents, lower throughput, smaller dynamic range, and higher risk for contamination, it facilitates a more sensitive molecular diagnosis and can be used for the absolute quantification under different conditions, such as absolute quantification without standard curve; moreover, it offers advantages such as lower contamination from sample inhibitors, reliable interlaboratory commutability, and better detection of low pathogen load (Kuypers and Jerome, 2017).
Overall, we established an RT-ddPCR assay for the detection and absolute quantification of ASGV without the standard curve. We optimized primer/probe concentrations and the annealing temperature, which increased the amplification efficiency and facilitated droplet separation. The optimized RT-ddPCR assay showed specificity for ACLSV, ASPV, and ASSVd, the major viruses that infect apple trees and are considered high-risk pathogens by KMAFRA while conducting virus-free certification programs. In addition, comparisons of RT-ddPCR with RT-qPCR showed that the sensitivity of the optimized RT-ddPCR assay was approximately 10-fold higher than that of RT-qPCR based on the serial dilution of ASGV-CP transcripts. For further evaluation of RT-ddPCR assay reliability, ASGV were detected and quantified in 37 micropropagated apple shoot samples. Of the 37 apple shoot samples, 35 samples (95%) tested positive for ASGV by RT-ddPCR analysis. These findings indicate that RT-ddPCR assay is more reliable than RT-PCR and RT-qPCR assays for detecting ASGV in apple shoot samples, even at low viral loads.
To the best of our knowledge, this is the first RT-ddPCR assay developed for the detection and quantification of ASGV infection. Our results indicate that this assay is suitable for ultra-sensitive and accurate diagnosis of ASGV infection. The successful detection of ASGV from several micropropagated apple shoot samples demonstrated that the RT-ddPCR assay can be used for routine surveys of ASGV infection and in screening ASGV infections while certifying ASGV-free apple plantlets.
Acknowledgments
This research was financially supported by Korea Institute of Planning & Evaluation for Technology in Food, Agriculture, Forestry & Fisheries (iPET) (320040-05-3-WT011).
Fig.┬Ā1
Thermal gradient reverse transcription droplet digital polymerase chain reaction (RT-ddPCR) for optimizing annealing temperature for the detection of apple stem grooving virus. Eight RT-ddPCR reactions (with an annealing temperature gradient ranging from 48┬░C to 62┬░C) were performed to optimize the annealing temperature. The horizontal pink line represents the threshold; positive droplets (blue) and negative droplets (gray) are shown above and below the line, respectively. The X and Y axes represent the number of droplets and fluorescence signal amplitude, respectively.
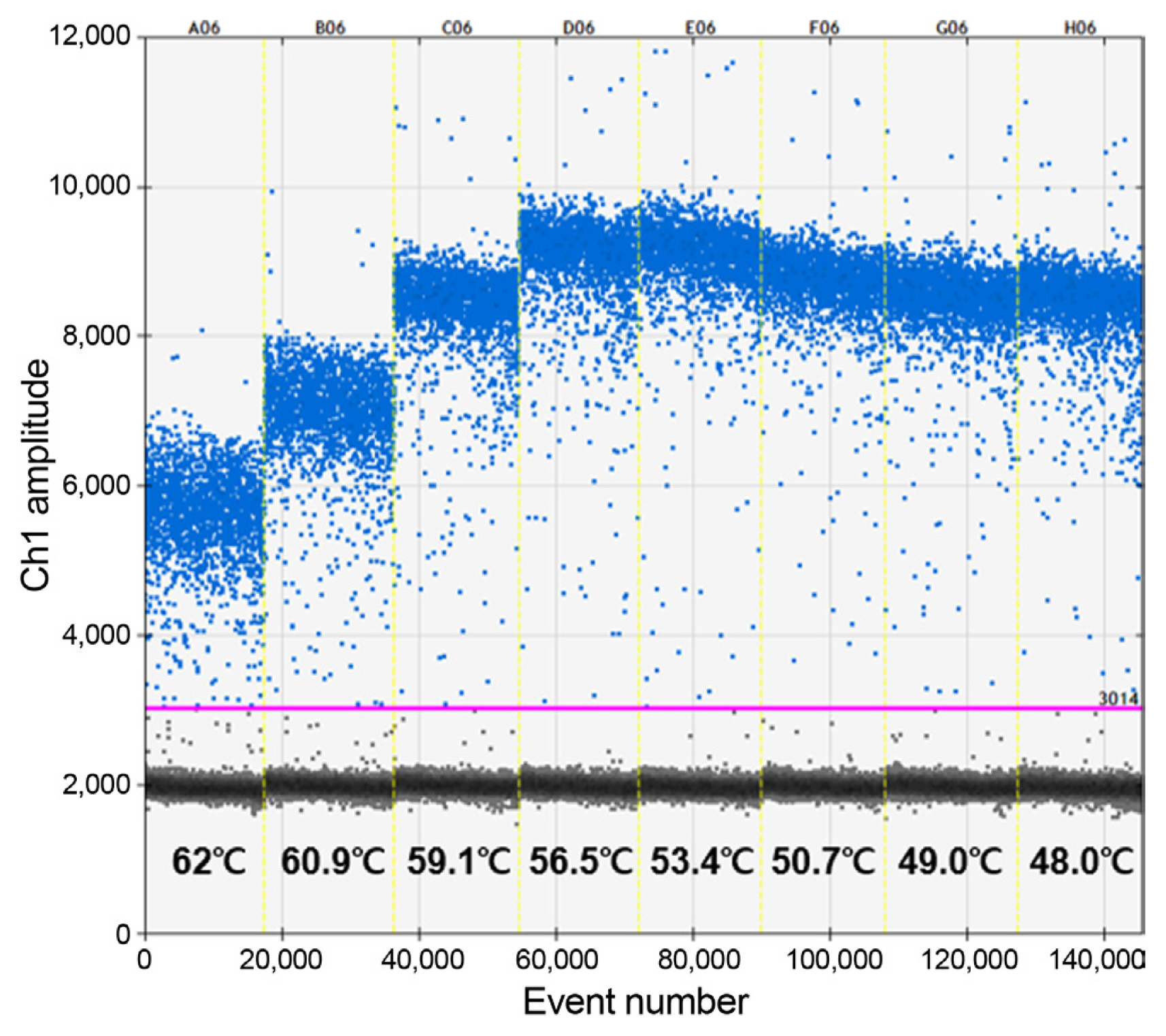
Fig.┬Ā2
Specificity of reverse transcription droplet digital polymerase chain reaction (assay for apple stem grooving virus (ASGV). Each sample is separated by vertical yellow lines and represents the fluorescence amplitude of apple chlorosis leaf spot virus (ACLSV), apple stem pitting virus (ASPV), apple skin scar viroid (ASSVd), and the negative template control (NTC).
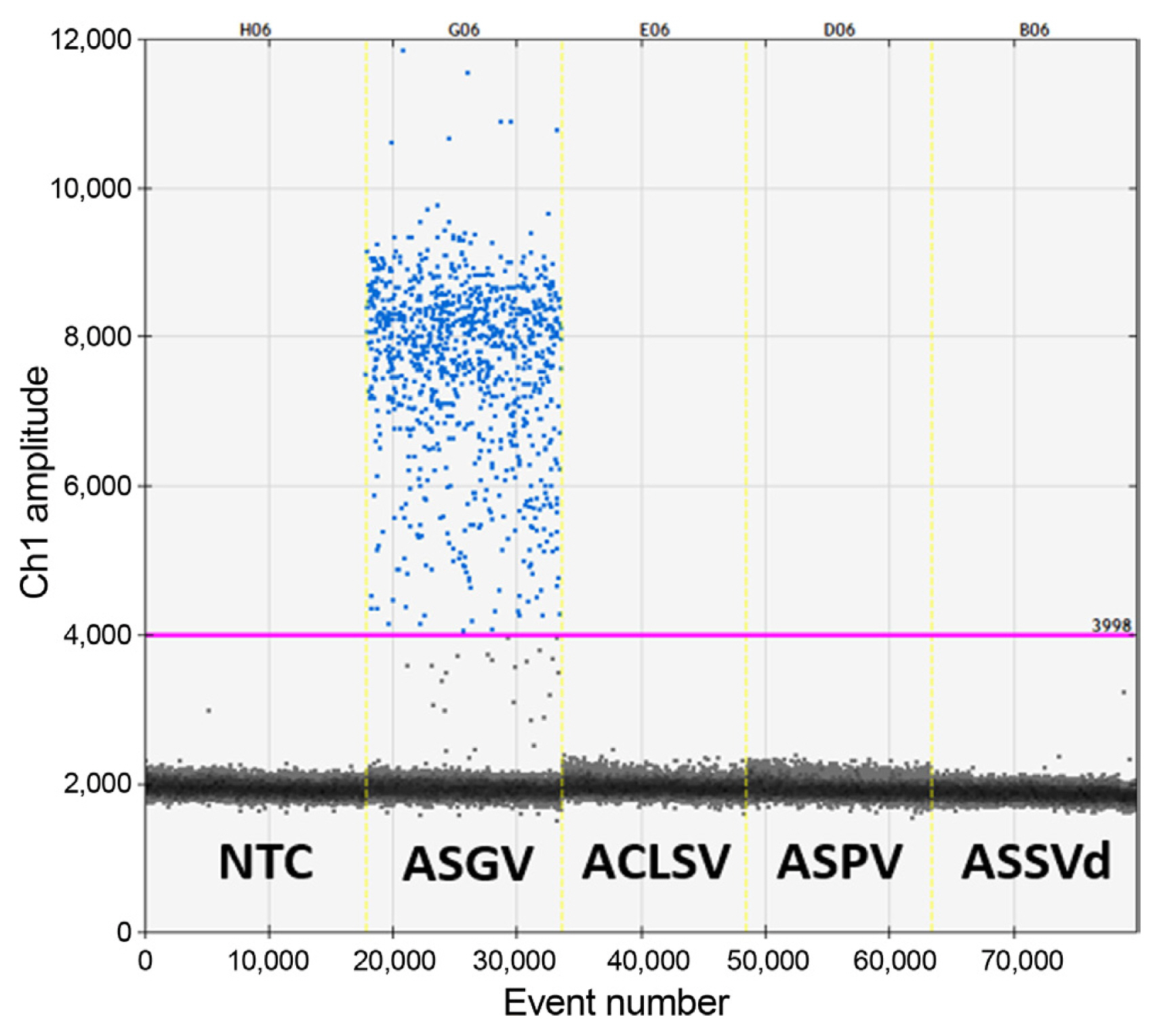
Fig.┬Ā3
Detection and absolute quantification of apple stem grooving virus in micropropagated apple plantlets by reverse transcription droplet digital polymerase chain reaction (RT-ddPCR). Lanes 1-3; non-template control (NTC), lanes 4-40; RNA samples from selected propagated apple plantlets. RT-ddPCR amplitude plot showing droplets with distinguished positives (mostly between 3,000 and 6,000) and negatives (mostly below 2,000) in 37 apple plantlet samples.

Table┬Ā1
Detailed information for the primers and probe used in this study
| Assay | Primer | Sequence 5ŌĆ▓ŌåÆ3ŌĆ▓ | Target gene | Position | Size (bp) | Reference |
|---|---|---|---|---|---|---|
| RT-qPCR and RT-ddPCR | ASGV-CNU-F | AGRCGCCACCGGGTAGG | ASGV CP | 5670-5686 | 100 | This study |
| ASGV-CNU-P | 6-FAM-ARVTTCTGACGGTTCCTCCCCCTGAA-BHQ-1 | ASGV CP | 5725-5750 | |||
| ASGV-CNU-R | CCTTCRAARCTTTCACCTTCTTTRA | ASGV CP | 5755-5779 | |||
| RT-PCR | ASGV-F | ATGAGTTTGGAAGACCTGCTTCAA | ASGV CP | 5640-5663 | 714 | Shim et al. (2006) |
| ASGV-R | CTAACCCTCCAGTTCCAAGTTACT | ASGV CP | 6330-6355 | |||
| Nad5-F | GATGCTTCTTGGGGCTTCTTGTT | Internal control | 1836-1859 | 181 | Menzel et al. (2002) | |
| Nad5-R | CTCCAGTCACCAACATTGGCATAA | 1973-1995 |
Table┬Ā2
Sensitivity of the RT-qPCR and RT-ddPCR assay for ASGV
Table┬Ā3
Concentration of ASGV in micropropagated apple plantlet samples by RT-PCR, RT-qPCR, and RT-ddPCR
References
Bhardwaj, P. and Hallen, V. 2019. Molecular evidence of apple stem grooving virus infecting Ficus palmata
. Trees 33:1-9.


Dube, S., Qin, J. and Ramakrishnan, R. 2008. Mathematical analysis of copy number variation in a DNA sample using digital PCR on a nanofluidic device. PLoS ONE 3:e2876.



Fronhoffs, S., Totzke, G., Stier, S., Wernert, N., Rothe, M., Br├╝ning, T., Koch, B., Sachinidis, A., Vetter, H. and Ko, Y. 2002. A method for the rapid construction of cRNA standard curves in quantitative real-time reverse transcription polymerase chain reaction. Mol. Cell. Probes 16:99-110.


Hindson, B.J., Ness, K.D., Maasquelier, D.A., Belgrader, P., Heredia, N.J., Makarewicz, A.J., Bright, I.J., Lucero, M.Y., Hiddessen, A.L., Legler, T.C., Kitano, T.K., Hodel, M.R., Petersen, J.F., Wyatt, P.W., Steenblock, E.R., Shah, P.H., Bousse, L.J., Troup, C.B., Mellen, J.C., Wittmann, D.K., Erndt, N.G., Cauley, T.H., Koehler, R.T., So, A.P., Dube, S., Rose, K.A., Montesclaros, L., Wang, S., Stumbo, D.P., Hodges, S.P., Romine, S., Milanovich, F.P., White, H.E., Regan, J.F., Karlin-Neumann, G.A., Hindson, C.M., Saxonov, S. and Colston, B.W. 2011. High-throughput droplet digital PCR system for absolute quantification of DNA copy number. Anal. Chem 83:8604-8610.



Hu, G.-J., Dong, Y.-F., Zhang, Z.-P., Fan, X.-D., Ren, F. and Li, Z.-N. 2018. Effect of pre-culture on virus elimination from in vitro apple by thermotherapy coupled with shoot tip culture. J. Integr. Agric 17:2015-2023.

Jeong, H.-W., Go, S.-M. and Jeong, R.-D. 2021. Rapid and specific detection of apple chlorotic leaf spot virus in pear by reverse-transcription recombinase polymerase amplification. Acta Virol 65:237-241.


Jiao, J., Kong, K., Han, J., Song, S., Bai, T., Song, C., Wang, M., Yan, Z., Zhang, H., Zhang, R., Feng, J. and Zheng, X. 2021. Field detection of multiple RNA viruses/viroids in apple using a CRISPR/Cas12a-based visual assay. Plant Biotechnol. J 19:394-405.




Kim, N.-Y., Lee, H.-J. and Jeong, R.-D. 2019. A portable detection assay for apple stem pitting virus using reverse transcription-recombinase polymerase amplification. J. Virol. Methods 274:113747.


Korean Statistical Information Service 2021 URL http://www.kosis.kr/index/index.do
. 30 March 2022.
Kuypers, J. and Jerome, K.R. 2017. Applications of digital PCR for clinical microbiology. J. Clin. Microbiol 55:1621-1628.




Lee, H.-J., Cho, I.-S., Ju, H.-J. and Jeong, R.-D. 2021. Development of a reverse transcription droplet digital PCR assay for sensitive detection of peach latent mosaic viroid. Mol. Cell. Probe 58:101746.

Lee, S., Cha, J.-S., Kwon, Y., Lee, Y.S., Yoo, S.E., Kim, J.H. and Kim, D. 2020. Occurrence status of five apple virus and viroid in Korea. Res. Plant Dis 26:95-102 (in Korean).


Lu, Y., Yao, B., Wang, G. and Hong, N. 2018. The detection of ACLSV and ASPV in pear by RT-LAMP assays. J. Virol. Methods 252:80-85.

Malandraki, I., Beris, D., Isaioglou, I., Olmos, A., Varveri, C. and Vassilakos, N. 2017. Simultaneous detection of three pome fruit tree viruses by one-step multiplex quantitative RT-PCR. PLoS ONE 12:e0180877.



Menzel, W., Jelkmann, W. and Maiss, D. 2002. Detection of four apple viruses by multiplex RT-PCR assay with coamplification of plant mRNA as internal control. J. Virol. Methods 99:81-92.

Nabi, S.U., Madhu, G.S., Rao, G.P. and Baranwal, V.K. 2022. Development of multiplex RT-PCR assay for simultaneous detection of four viruses infecting apple (Malus domestica). Lett. Appl. Microbiol 74:586-592.



Pfaffl, M.W. and Hageleit, M. 2001. Validities of mRNA quantification using recombinant RNA and recombinant DNA external calibration curves in real-time RT-PCR. Biotechnol. Lett 23:275-282.
- TOOLS
-
METRICS

- ORCID iDs
-
Rae-Dong Jeong

https://orcid.org/0000-0002-6370-8909 - Related articles



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Supplement1
Supplement1 Print
Print



