Antifungal Effect of Chitosan as Ca2+ Channel Blocker
Article information
Abstract
The aim of this study was to investigate antifungal activity of a range of different molecular weight (MW) chitosan against Penicillium italicum. Our results demonstrate that the antifungal activity was dependent both the MW and concentration of the chitosan. Among a series of chitosan derived from the hydrolysis of high MW chitosan, the fractions containing various sizes of chitosan ranging from 3 to 15 glucosamine units named as chitooligomers-F2 (CO-F2) was found to show the highest antifungal activity against P. italicum. Furthermore, the effect of CO-F2 toward this fungus was significantly reduced in the presence of Ca2+, whereas its effect was recovered by ethylenediaminetetraacetic acid, suggesting that the CO-F2 acts via disruption of Ca2+ gradient required for survival of the fungus. Our results suggest that CO-F2 may serve as potential compounds to develop alternatives to synthetic fungicides for the control of the postharvest diseases.
Introduction
Fresh fruits like citrus are very susceptible to postharvest decay which decrease their storage periods, values and marketing life. This postharvest decay of fresh fruits results in considerable economic losses in industry production of citrus fruits (Zhang et al., 2011). Citrus fruits cultivation is very important in China, the largest producer of the citrus fruits in the world. However, estimated losses of citrus fruits due to blue mold infection by Penicillium digitatum and/or Penicillium italicum are known to be up 40% in China (Sun et al., 2011). Hence, synthetic fungicides (SFs) including sodium ortho-phenlyphenol, pyrimethanil and imazalil have long been commonly used as method for controlling postharvest disease of citrus fruits (Lahlali et al., 2005; Yoshioka et al., 2004). However, this indiscriminate use of the SFs on citrus fruit cultivation has several disadvantages such as potential harmful effects on human health, the development of pathogen resistance toward fungicidal chemicals and environmental ecosystem (Al-Samarrai et al., 2012). Therefore, the need for seeking natural pollution-free sources toward the development of new alternatives, which pose little or no risk to human health is in urgent demand (Ortelli et al., 2005).
The search for chitosan as a natural alternative among natural bioactive substances for controlling postharvest diseases has been evaluated (Badawy et al., 2004). Chitosan is produced from deacetylation of chitin, which is usually obtained from shells of krill, shrimp, crab, insect cuticle and mushroom cell wall (Kaya et al., 2015a, 2015b). It consists of -(1,4)-2-acetamido-2-deoxy-Dglucose and -(1,4)-amino-2-dexoy-D-glucose units. Regarding the primary structure of chitosan, its polycationic nature allows this polymer to exert numerous and unique biological functions with great potential applicability in various industries including agriculture, medicine and pharmacology (Badawy et al., 2005; Bhaskara Reddy et al., 1999; Tayel et al., 2010; Wojdyła, 2004). In particular, the antifungal effects of chitosan and its derivatives have attracted considerable attention for several decades. All types and concentration of chitosan gave the most potential inhibitory effect on mycelia growth in P. digitatum and P. italicum (Cháfer et al., 2012; Muñoz and Moret, 2010; Tayel et al., 2016; Vitalini et al., 2014). Other studies found no differences in the fungicidal pattern among different types of chitosan. In contrast, their fungicidal effect increased by incorporation of chitosan resulted in a great reduction in fungal growth and viability (Tayel et al., 2016). Furthermore, it has been reported that chitosan showed outstanding effectiveness in inhibiting spore germination, germ tube elongation and radial growth of Botrytis cinerea or Rhodosporidium paludigenum, indicating that this polymer induced morphological alterations depending on MW and/or the degree of deacetylation or alkylation of chitosan (Lahlali et al., 2005; Lu et al., 2014; Mazur and Waksmundzka, 2001; Rabea et al., 2006; Younes et al., 2014). In general, it has been well known that the antibacterial and antimicrobial activity of chitosan are due to its polycationic nature that can cause the leakage of intracellular electrolytes and proteinaceous constituents or enter fungal cells and then inhibit or slow down the synthesis of messenger RNA (mRNA) and protein (Hadwiger, 2013; Palma-Guerrero et al., 2010; Tayel et al., 2010; Tsyhankova et al., 2012). Despite some progress, achieved in the past few years, the complete action modes of chitosan with different molecular weight (MW) including the degree of deacetylation (DD) of chitosan towards P. italicum still remain unclear. Therefore, the present study aimed to determine the synergistic effect of chitosan with a range of different MW on the antifungal activity, using chitosan designated to high MW chitosan (HMWC) and medium MW chitosan (CO-F2) in vitro morphological development such as mycelia growth and germination spores of P. italicum. In order to reduce the burden of working time, both MTT [3-(4, 5-dimethylthiazol- 2-yl)-2, 5-diphenyltetrazolium bromide] and WST [(2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium)] reduction assays were performed for quantitative measurement of the synergistic effect of chitosan in the presence or absence of antibiotics. Furthermore, we demonstrated for the first time, to the best of our knowledge, that the antifungal effects of the CO-F2 is the result of the disturbance of Ca2+ gradients, thereby revealing a previously unknown role for CO-F2 as fungal Ca2+ channel blocker.
Materials and Methods
Materials
HMWC with approximately 2,000 kDa, glucosamine hydrochloride (GlcN) and N-acetyl glucosamine (GlcNAc) were purchased from Sigma Chemical Co. (St. Louis, MO, USA). Trypan blue and chitosanase from Streptomyces griseus were purchased from Sigma Chemical Co. Potato dextrose broth (PD) was purchased from Difco™ (Becton Dickinson, Cockeysville, MD, USA). MTT, WST, thiabendazole and imazalil were also purchased from Sigma Chemical Co. All other reagents were used without further purification and were of the highest grade available.
Preparation of CO-F2
The HMWC was fully dissolved in 2% acetic acid to make 2% (w/v) solution (100 ml), incubated at 37°C for 1 hour to warm up the solution before add the enzyme. In order to hydrolyze the HMWC, the chitosanase (1.0 U/ml) was added into the solution of HMWC. Chitosanase activity was determined at 37°C in a time dependent manner by measuring the concentration of reducing sugars derived from the hydrolysis of HMWC using PAHBAH saline (4-hydroxy-benzhydrazid), spectrophotometrically. In order to determine the optimal temperature for the production of CO-F2, enzyme reaction was carried out at a wide range of temperatures from 20°C to 60°C. The process was performed under the optimal temperature of free enzyme, after which the reaction was quenched by boiling for 5 minutes at 100°C, cooled down on ice for 10 minutes and centrifuged at 13,500 rpm for 30 minutes to remove denatured enzyme and undigested materials. Of note, 98% of ethanol was added to precipitate CO-F2, since products smaller than 0.5-kDa of MW of chitosan which was produced from the hydrolysis of HMWC can easily not separate by ethanol precipitation. The precipitates were then washed with the ethanol until the pH value of the CO-F2 solution was same as the ethanol. After washing with ethanol several times, the final products were dried by using a lyophilizer (Il-Shin, Dongducheon, Korea).
Instrumental analysis
The CO-F2s with different MW obtained from the hydrolysis of HMWC were applied to high performance liquid chromatography (HPLC) and/or matrix-assisted laser desorption/ionization-time of flight mass spectrometry (MALDI-TOF MS, Voyager-DE™STR Biospectrometry Workstation; Applied Biosystems Inc., NCIRF, Seoul, Korea) analysis to obtain the pattern of the size of CO-F2. In the HPLC analysis, the MW of CO-F2 was calculated from calibration curve and area ratio of major peaks. In the MALDI-TOF MS, average MW of the CO-F2 was calculated based on the single unit of GlcN and/or GlcNAc for hetero chitooligomers.
Antifungal activity of the CO-F2
Fungi P. italicum (1 × 105/ml) was grown at 25°C in a sterile 96-well plate in a final volume of 200-μl PD medium (2.4% PD broth in distilled water) in the presence or absence of HMWC or CO-F2 at different concentrations ranging from 0 to 50 μg/ml under asepsis conditions. The growth was determined by measuring the absorbance at 495 nm microscopically and morphological alteration of the P. italicum was observed by trypan blue staining. Briefly, 50-μl of trypan blue solution (0.5% trypan blue in 45% acetic acid) was loaded into the test wells containing the mixture of P. italicum and each sample of 0.005% (w/v) as the final concentration. After reaction for 5 minutes at room temperature, microscopic observation was performed. In all the experiments, each treatment was performed with triplicate samples, and the mean ± standard deviation (SD) were calculated for each sample.
MTT and WST reduction assay
A relative viability of P. italicum affected by HMWC, CO-F2 and commercialized fungicides such as thiabendazole and imazalil was evaluated by using a modified MTT and/or WST-reduction assay (Fisichella et al., 2009; Ngamwongsatit et al., 2008). Briefly, MTT and WST were prepared in PD medium to be 5 mg/ml. Then each solution was filtered through a 0.22-μm pore size filter. Prior to each assay, P. italicum was cultured in the presence or absence of the chitosan samples for 3 days at 30°C. After the cultivation, 0.5 mg/ ml of MTT or WST solution was added to each well. The mixtures were then incubated in a dark room for 2 hours at room temperature. For MTT-reduction assay, formazan-crystal produced by reductase was dissolved by dimethylsulfoxide. A colorimetric change in the MTT- or WST-reduction assay was measured spectrophotometrically using micro-plate reader (uQuant; Bio-Tek, Seoul, Korea) at 570 nm or 450 nm, respectively, and the relative viability was calculated by comparing with the absorbance change of the control. The minimum inhibitory concentration (MIC) was defined as the lowest concentration of samples that showed no growth observed in the culture medium at the end of the experiments.
Effects of divalent cations on CO-F2 activity
The inhibitory activities of CO-F2 in the presence of divalent cations (Ca2+, Mg2+, Zn2+, and Mn2+) on fungal spore germination were determined by micro-spectrophotometry of liquid cultures as previously described (Koo et al., 1998). Briefly, PD medium (0.1 ml) containing 104 fungal spores per well in a microplate in the presence or absence of CO-F2 and divalent cations was prepared, respectively. After incubation at 24°C for 2 days without shaking, the optical density of the cultures was measured using a microplate reader (Infinite M200 Proquant; Tecan, Salzburg, Austria) at 600 nm. The MIC 50% (MIC50) was determined from dose-response curves.
For the statistical analysis of data, all results obtained from three separate independent experiments were expressed as the means ± SD, and were considered as statistical significances at P < 0.05.
Results and Discussion
In this study, we characterized the anti-fungal effects of chitosan. We prepared a range of different MW chitosan by HMWC hydrolysis. In order to further separate active size of chitosan, total hydrolysates were fractionated depending on the size of molecules using filter membranes ranging from 3- to 10-kDa cut-off. Among four fractions, fraction-2 designated as CO-F2, revealed the highest anti-fungal activity, and thus was further analyzed using HPLC to determine the average MW. The average MW of CO-F2 was determined to be 2.1-kDa by HPLC analysis. Despite our inability to complete the portion ratio of each molecule we were able to obtain some insight into the distribution of various sizes of molecules. From six to thirteen units of multimeric glucosamines in CO-F2 were determined to be the major products appearing as the major peak during the monitoring, and were subsequently identified by using MALDI-TOF MS analysis (Fig. 1). It has been recognized, for several decades now, that the biological effects of chitosan include antifungal activity depending on its MW and DD, pH of chitosan solution and type of target organism (Kendra and Hadwiger, 1984). Although several mechanisms were proposed for the antifungal activity of chitosan especially (Rabea et al., 2006; Tayel et al., 2016; Tsyhankova et al., 2012; Zahid et al., 2012), mainly effects on fungal cell wall, precise mechanism of the antifungal activity of chitosan is still under the investigation.
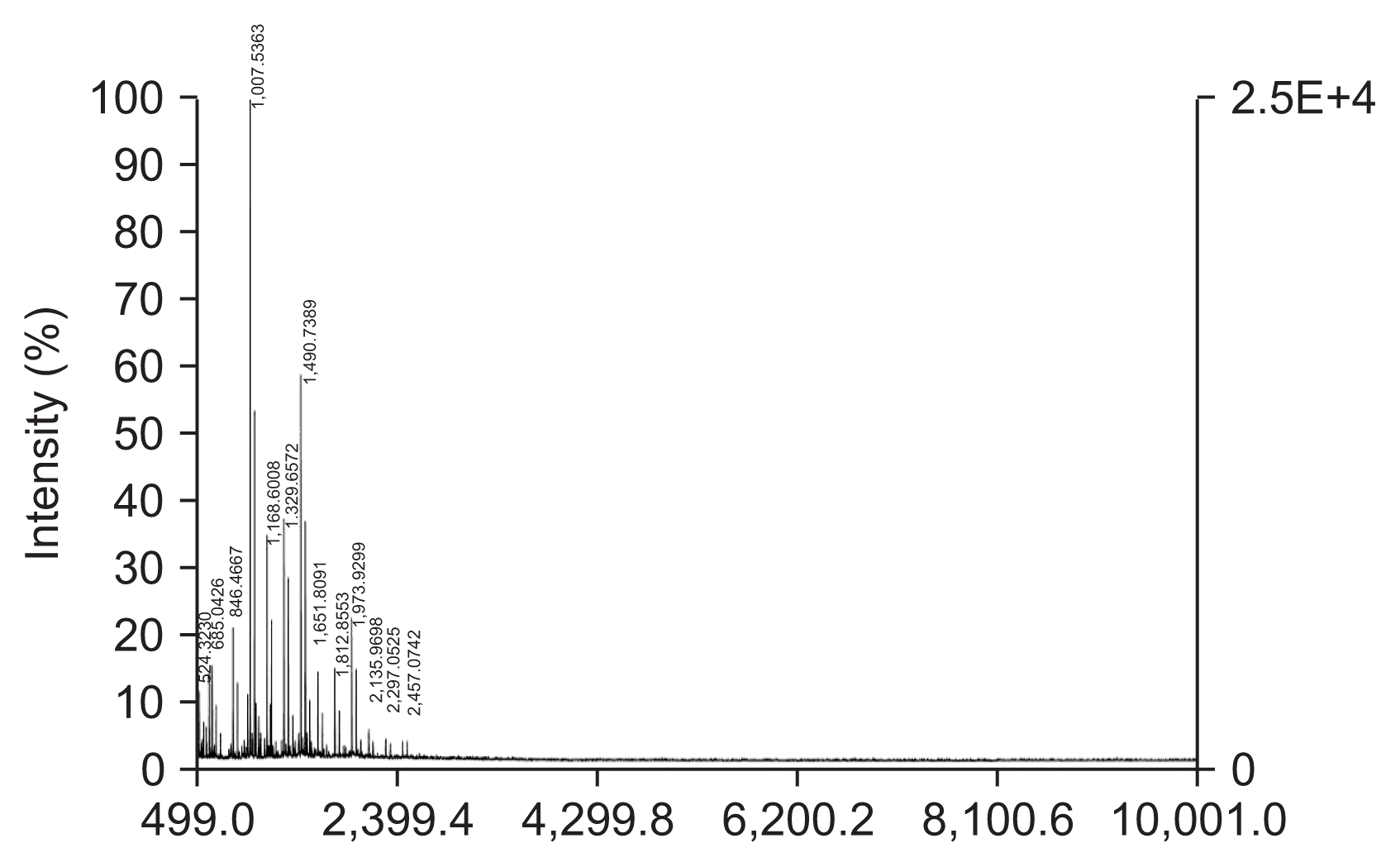
MALDI-TOF mass analysis of chitooligomers-F2 (CO-F2). CO-F2 consisting of (GlcN)n, n = 3–15 as major products were analyzed and identified in full scanned scale ranging from 499 to 10,001 m/z by MALDI-TOF mass spectrometry.
Antifungal activity of HMWC and CO-F2 derived from the hydrolysis of HMWC was investigated against P. italicum. As shown in Fig. 2, CO-F2 exerted stronger antifungal activity than that of HMWC, thus suggesting that its short chain possess more efficient inhibitory action on fungal growth (Fig. 2D). As described in the introduction section, chitosan may cause the leakage of cell wall using their polycationic nature or enter fungal cells, and then inhibit or slow down the synthesis of mRNA and protein. It is reasonable to speculate that CO-F2 could exert its antifungal effect through multiple mechanisms towards due to its short chain capable of disturbing fungal cell wall, which is agreement with previous reports (Cohen, 1993). Quaternized chitosan was described to act on the fungal cell surface because as a result of its large size. Herein, we found the antifungal effects of HMWC and CO-F2 to be concentration dependent. Of note, the most potent antifungal activity of CO-F2 was observed at 50 μg/ml. Interestingly, the antifungal effects of CO-F2 on P. italicum exceeded those of HMWC (Fig. 2C). This is probably related to the lower binding capability of HMWC toward the structural components of the cell wall of P. italicum as compared with CO-F2. Although our data demonstrate a lack of correlation between MW of chitosan and its binding capacity to the cell wall, a relevant finding is that CO-F2 is capable of inhibiting the growth of P. italicum. We also confirmed that exogenously added Ca2+ and Mg2+ (5 nM) did not significantly inhibit the growth of the fungus in low concentration dependent manner (Fig. 2E, F), respectively, although ethylenediaminetetraacetic acid (EDTA; 5 nM) had a slight effect on the mycelium growth (Fig. 2B). Therefore, our results demonstrate that the underlying antifungal mechanisms are independent of each molecule, in order to attain more effective protection.
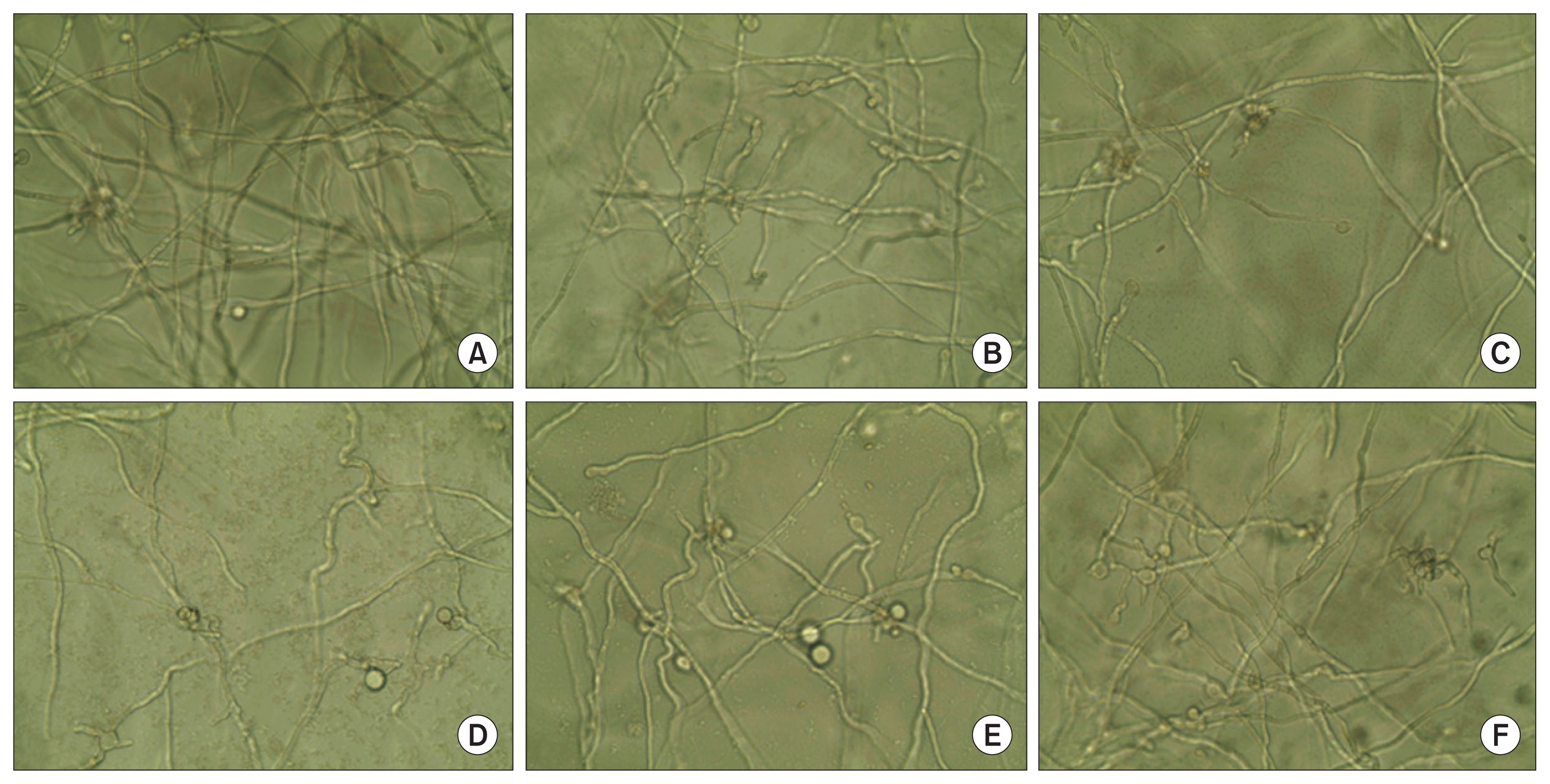
Morphological alteration of Penicillium italicum. P. italicum was incubated in the presence of chemical compounds including high molecular weight chitosan (HMWC) and chitooligomers-F2 (CO-F2) in potato dextrose (PD) culture medium. After 3 days of incubation at room temperature, morphological changing of P. italicum was observed under a microscope (× 400). (A) Control. (B) 5 nM ethylenediaminetetraacetic acid. (C) 10 μg/ml of HMWC. (D) 10 μg/ml of CO-F2. (E) 5 nM Ca2+. (F) 5 nM Mg2+.
Since cytosolic Ca2+ gradient in fungus is critical for survival under various environmental stresses such as hypo- and hyper-osmotic shock (Brand et al., 2009; Miller et al., 1990; Silverman-Gavrila and Lew, 2001). For instance, Ca2+ is involved in hyphal elongation (Brand et al., 2007; Jackson and Heath, 1993; Levina et al., 1995), bud formation and cyclic adenosine monophosphate regulation (Bencina et al., 2005). Ca2+ gradient in fungi is maintained through a series of Ca2+ channels. This prompted us to investigate influence of Ca2+ on the antifungal activity of the CO-F2. We observed reduced antifungal activity of the CO-F2 upon the addition of exogenous 5 nM Ca2+ (Fig. 3D), indicating that Ca2+ is essential for CO-F2 antifungal activity (Fig. 3E). Collectively, we suggest that the CO-F2 may act via disruption of a Ca2+ gradient or balance required for survival and further blocking specific fungal Ca2+ channel (Brand et al., 2009) (Fig. 3C). This finding was confirmed again upon evaluating the inhibitory activity of CO-F2 on fungal growth, thus demonstrating the antagonistic effect of Ca2+ in a concentration dependent manner.

Trypan blue staining of Penicillium italicum. Morphological alteration of P. italicum was observed by staining using trypan blue solution (0.005% trypan blue/0.45% acetic acid) in the presence of chemical compounds to elucidate the synergistic effect of chitooligomers-F2 (CO-F2). (A) Control. (B) 5 nM of ethylenediaminetetraacetic acid (EDTA). (C) 10 μg/ml of CO-F2. (D) 5 nm of EDTA + 5 nM of Ca2+. (E) 10 μg/ml of CO-F2 + 5 nM of Ca2+.
MTT or WST assay is relevant to mitochondrial reductase in the live cells. This reductase can convert a tetrazolium series including MTT and WST into a reduced formazan-colored product which could be measured spectrophotometrically. These assays were used to quantitatively measure the antifungal effect of chitosan samples. We found CO-F2 (about 40–50%) to show stronger antifungal activity than that of HMWC (about 20–30%) (Fig. 4A), which is consistent with the results of microscopic observation (Fig. 2). Moreover, the antifungal activity of CO-F2 increased with its concentrations from 0 to 25 μg/ml (Fig. 4B). Of note, a fungicidal effect for chitosan with different MW, which affected spore germination and hyphae morphology, was previously observed and reported at (1.0–2.0 mg/ml) (Olicón-Hernández et al., 2015; Robles-Martínez et al., 2014). This significant difference may be due to agglomerative feature of chitosan, which suggests that the mode of chitosan antifungal action was inhibited by agglutinates consisting of PD and chitosan. This is likely a factor during microscopic observation. Therefore, it is essential to take into account the effect of culture medium used and its influence on the antifungal activity of chitosan.
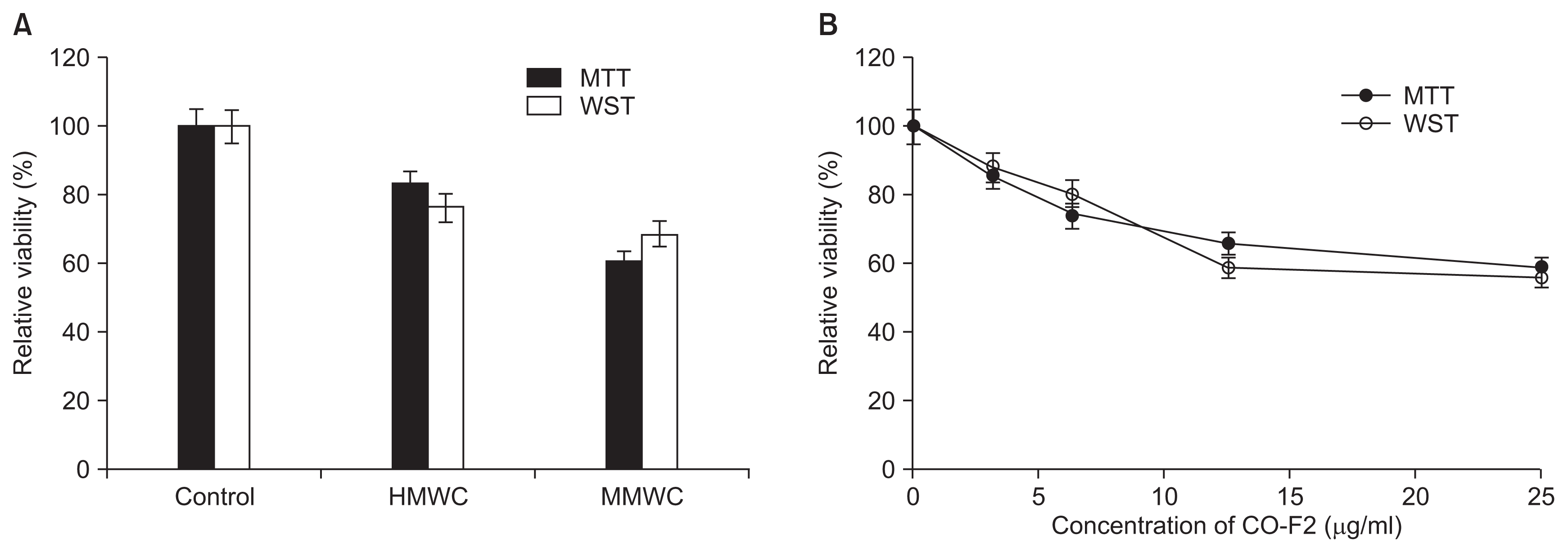
MTT and WST reduction assay. Values represent the mean and standard deviations for the multiple independent formazan dye production. (A) In experiment A, each sample was tested in the presence of chitosan series (10 μg/ml) or absence of sample (control), respectively. (B) In experiment B, relative viability of fungi was tested with different concentration of CO-F2 ranging from 0 to 25 μg/ml. MTT, [3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide]; WST, [(2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium)]; CO-F2, chitooligomers-F2; HMWC, high molecular weight chitosan; MMWC, medium molecular weight chitosan.
There exists the need for natural fungicide with no toxicity, especially with the wide spread resistance of P. italicum to SFs currently used in agricultural field. Therefore, in this study, the antifungal activity of the CO-F2 and SFs such as thiabendazole and imazalil was compared to evaluate whether the CO-F2 could be used as attractive alternative for the currently used SFs. The result showed that the P. italicum was more susceptible to thiabendazole (40%) than imazalil (20%) and the CO-F2 (~50%) tested exerted stronger activity than both SFs (Fig. 5). These results suggest that the CO-F2 acts more efficiently than the SFs. Although, HMWC showed less activity by ~10% than that of the SFs, it could be used for edible plants such as vegetables and fruits as alternatives to SFs, since it has no detectable toxicity, in addition to its biocompatible and biodegradable properties. Interestingly, we observed some variation between the results of Fig. 4 and 5, showing that activity of the HMWC was slightly lower in result of Fig. 5 than that of in Fig. 4. These results displayed drastic deviation especially in WST assay. Possible explanations for the observed variations in antifungal activity of the chitosan samples used in this study include differences in fungal cell wall absorption, interaction with the molecules in cell wall or membrane penetration abilities of chitosan samples, as well as different binding capability of the different MW of chitosan samples toward the structural components of the cell wall of P. italicum.

Comparison of the antifungal activity of CO-F2 with fungicides. Chitosan series of HMWC and CO-F2, and fungicides (10 μg/ml) were used for the determination of antifungal activity, respectively. Relative viability of fungi was determined by MTT and WST reduction assay as described in the materials and methods section. MTT, [3-(4, 5-dimethylthiazol- 2-yl)-2, 5-diphenyltetrazolium bromide]; WST, [(2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium)]; HMWC, high molecular weight chitosan; CO-F2, chitooligomers-F2.
Effect of divalent cations on CO-F2 activity
In order to determine the availability of essential metal ions required for the integrity and viability of P. italicum, we examined the effect of physiologically abundant divalent cations such as Ca2+, Mg2+, Mn2+, and Zn2+ on CO-F2’s antifungal activity. A number of divalent cations have been implicated as important biological factors in controlling cellular responses and cell wall biogenesis, including those involved in environmental stresses loaded to fungal cells. Our results show that the addition of Mg2+, Mn2+, and Zn2+ to the assay mixture did not influence CO-F’s activity against CO-F2. Moreover, there was no significant (P > 0.05) change in the MIC50 values were observed compared to those of non-treated controls (data not shown). However, we observed a significant dose-dependent reduction of CO-F2’s activity upon addition of Ca2+ (Fig. 6A). Earlier studies demonstrated that the activation of the calcineurin and PKC1 pathway via the addition of Ca2+ causes cell growth arrest as well as defective cell lysis in response to cell wall stress in yeast (LaFayette et al., 2010; Levin et al., 1990), indeed Ca2+ has antogonistic effect on CO-F2’s fungal growth inhibitory activity. In contrast, CO-F2’s fungal growth inhibitory activity was gradually recovered by addition of EDTA. This result demonstrated that EDTA combines with Ca2+ and suppresses its ability to bind CO-F2 thus releasing CO-F2, which in turn can exert its inhibitory effects on fungal growth (Fig. 6B). The MIC50 value of CO-F2 toward P. italicum was determined to be about 40 μg/ml. Interestingly, our data exhibited similar inhibitory activity against twenty strains of Fusarium fujikuroi isolated in South Korea (unpublished data), obtained from the Korean Agricultural Culture Collection. Based on the results, we suggest that fungal growth inhibitory activity of CO-F2 might be mediated through interactions with specific molecules related to Ca2+ homeostasis in P. italicum.
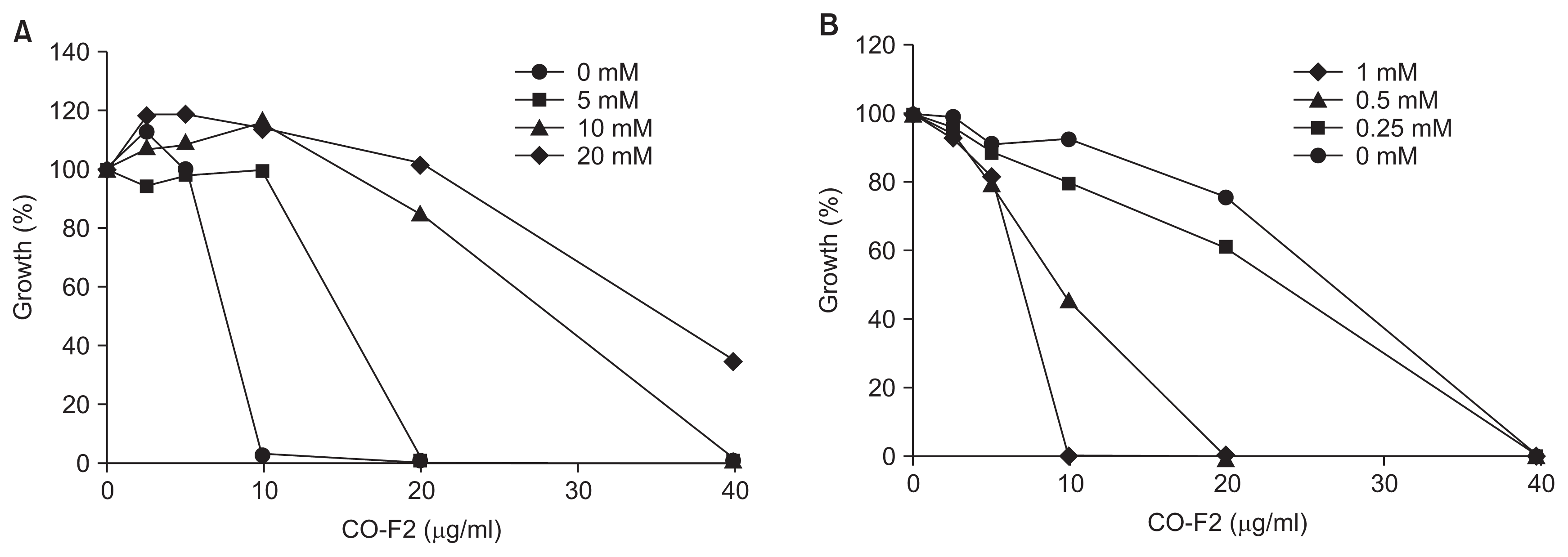
Antagonistic effect of Ca2+ on CO-F2’s activity. Calcium ions ranging from o to 20 mM were used for the determination of antagonistic effect on the inhibitory growth effect of chitooligomers-F2 (CO-F2), in the absence (A) or presence (B) of ethylenediaminetetraacetic acid (0–1.0 mM), respectively. Relative growth of fungi was determined by micro-spectrophotometry of liquid cultures as described in the materials and methods section.
In conclusion, the antifungal activity of chitosan against P. italicum was characterized and found to be MW and concentration dependent. Our results showed that the CO-F2 (2.1 kDa) is more effective in inhibiting fungal growth than HMWC. Furthermore, we found the antifungal effect of CO-F2 to become reduced in response to exogenously added Ca2+, suggesting that the CO-F2 tested acts via disruption of a Ca2+ gradient and further may serve as Ca2+ channel blocker. These results suggest that multiple mechanisms are likely to drive the antifungal action of chitosan, which may include the inhibition of mRNA or protein synthesis. Further studies are required to elucidate the precise chitosan’s mode of action on P. italicum and other fungi. Overall, our results provide some evidence to support the investigation and development of fungicidal compounds as alternatives to commercial fungicides for controlling the postharvest diseases caused by P. italicum and other related fungi.
Supplementary Information
Acknowledgments
This work was supported by the research foundation (GCU-2015-0057) of the Gachon University, Korea. The authors also would like to express sincere thanks to Dr. Mawadda Alnaeeli, Division of Infectious Diseases, School of Medicine, University of Louisville, Louisville, KY 40202, USA, for valuable comments and edition for this study.
Notes
Articles can be freely viewed online at www.ppjonline.org.