 |
 |
| Plant Pathol J > Volume 32(3); 2016 > Article |
Abstract
The present study investigated the suppression of the disease development of anthracnose caused by Colletotrichum gloeosporioides and C. acutatum in harvested apples using an antagonistic rhizobacterium Paenibacillus polymyxa APEC128 (APEC128). Out of 30 bacterial isolates from apple rhizosphere screened for antagonistic activity, the most effective strain was APEC128 as inferred from the size of the inhibition zone. This strain showed a greater growth in brain-heart infusion (BHI) broth compared to other growth media. There was a reduction in anthracnose symptoms caused by the two fungal pathogens in harvested apples after their treatment with APEC128 in comparison with non-treated control. This effect is explained by the increased production of protease and amylase by APEC128, which might have inhibited mycelial growth. In apples treated with different APEC128 suspensions, the disease caused by C. gloeosporioides and C. acutatum was greatly suppressed (by 83.6% and 79%, respectively) in treatments with the concentration of 1 × 108 colony forming units (cfu)/ml compared to other lower dosages, suggesting that the suppression of anthracnose development on harvested apples is dose-dependent. These results indicated that APEC128 is one of the promising agents in the biocontrol of apple anthracnose, which might help to increase the shelf-life of apple fruit during the post-harvest period.
Anthracnose is one of the important post-harvest diseases of apple fruit caused by Colletotrichum gloeosporioides and C. acutatum, resulting in serious damage to the fruits and considerable economic loss in various crops, including apple worldwide (Afanador-Kafuri et al., 2003; Bajpai et al., 2009; Lee et al., 2007; Víchová et al., 2012). The symptoms of apple anthracnose can be characterized as follows: the lesions begin as a small, light brown, circular lesion, and later gradually expands. As the lesion expands, conidiophores rupture the fruit epidermis, forming small tufts (Henz et al., 1992). The fruit decays rapidly as it ripens and eventually become mummified that may remain attached to the tree throughout the season. Under humid conditions, large numbers of spores are produced in a creamy mass, while under dry conditions, the spore mass appears crystalline. The rotted flesh beneath the surface of the lesion is watery, appearing in a V-shaped pattern in cross section that narrows toward the core (Onofre and Antoniazzi, 2014). They invade the plant tissues aggressively, killing the host cells to obtain nutrients (Deacon, 1997).
The occurrence of anthracnose and other post-harvest diseases of fruits have been controlled mostly by the use of chemical treatments. Biological control of plant diseases using microorganisms has been an effective alternative to chemical control for a long time, and many biocontrol agents have been introduced in the past several years to control fruit diseases, thus reducing the regular use of fungicides (Mari et al., 2009). Biocontrol of post-harvest diseases is dependent on a quantitative relationship between the antagonist and the biocontrol agent (Nunes et al., 2002).
The antagonistic microorganisms, including both fungi and bacteria have been extensively studied as biocontrol agents against various soil-borne pathogens (Cook, 2000). Among the 20 genera of bacteria, Bacillus spp., Pseudomonas spp., and Streptomyces spp., are widely used as biocontrol agents. Bacillus spp., produced different kinds antibiotics (Ferreira et al., 1991). The genus Paenibacillus is comprised of more than 30 species of facultative anaerobes and belongs to endospore-forming gram-positive bacilli. Paenibacillus polymyxa, which was referred to as Bacillus polymyxa until 1993 (Ash et al., 1993), is a soil bacterium from the group of plant growth-promoting rhizobacteria, being used for the biocontrol of plant diseases (Dijksterhuis et al., 1999; Shishido et al., 1996). The activities associated with P. polymyxa treated plants in the earlier reports include nitrogen fixation (Heulin et al., 1994), soil phosphorus solubilization (Singh and Singh, 1993), and production of antibiotics (Rosado and Seldin, 1993), chitinase activity (Mavingui and Heulin, 1994), and the promotion of increased soil porosity (Gouzou et al., 1993). All these activities contributed to the promotion of plant growth at various times under various environmental conditions during the life cycle of a plant.
Until now, no information on the effectiveness of P. polymyxa strain on the control of post-harvest diseases in apple has been reported. Therefore, the main objective of this study was to investigate the efficacy of the strain P. polymyxa APEC128 in the control of anthracnose disease on apples after harvest.
Soil samples (50 g) were collected from several orchard apple trees free from disease symptoms in Andong, Gyeongbuk Province, South Korea to isolate antagonistic bacteria against apple pathogens. The soil samples were stored at 4°C in sterile plastic bags until their use. Soil samples (5 g) were diluted with 45 ml of sterile distilled water (SDW) and thoroughly dispersed by shaking at 150 rpm for 30 minutes at 28°C and from this 1 ml is taken and diluted 103- to 107-fold. Diluted samples (100 μl) were spread on nutrient agar (NA) medium (Kloepper, 1992), and incubated at 28°C for 24 hours. A single colony from each plate was picked up and streaked on freshly prepared NA plates. A total of 30 isolates were selected randomly from the culture plates, and the purified strains were stored on NA at 4°C. The isolated strains were maintained at −50°C in nutrient broth (NB) with glycerol (20%) for long-term storage. For preparing bacterial suspensions, a culture stored at −50°C was grown on NA for 24 hours at 28°C, and single colonies were transferred to NB and incubated for 24 hours at 28°C with shaking at 150 rpm. Bacteria were pelleted after centrifugation for 5 minutes at 8,000 rpm and resuspended in SDW to give a final concentration of 1 × 108 colony forming units (cfu)/ ml (OD600 nm) before application.
All the microbial isolates were tested in vitro for antagonistic effect against C. gloeosporioides (KACC 42390) and C. acutatum (KACC 40847) on potato dextrose agar (PDA; Difco, Detroit, MI, USA) including 0.5% peptone (PDAP) using a disc diffusion method (Park et al., 2013). The tested fungus (C. gloeosporioides or C. acutatum) was applied as mycelia plugs (6 mm in diameter) from the edge of a pre-cultured colony and placed onto the center of each PDAP plate (90 mm diameter). A sterile paper disk (8 mm diameter; Advantec Co., Tokyo, Japan) with a bacterial suspension (108 cfu/ml) was placed 30 mm from the pathogenic fungi. The inhibition of the mycelial growth of tested fungus was measured as a diameter (mm) 5 days after incubation at 25°C. The organism that produced the largest zone of inhibition during in vitro screening was selected for further study. Out of 30 strains screened, only one strain (APEC128) showed strong antagonistic activity and was selected for further experiments in this study. Each treatment contained five replications, and the assay was repeated three times, and results of one of the three experiments are shown here.
Fungal pathogens C. gloeosporioides (KACC 42390) and C. acutatum (KACC 40847) were obtained from Korean Agricultural Culture Collection. Formerly, the pathogens were isolated from symptomatic apple fruits as follows: Symptomatic tissues were surface sterilized in 1% sodium hypochlorite (NaOCl) solution for 1 minute and then placed in 70% ethanol for 30 seconds, followed by rinsing twice in SDW. After sterilization, the tissues were dried on sterile filter paper and transferred onto PDA plates. The plates were incubated at 25°C for 7 days. Single conidia were obtained by spreading PDA plates with conidia suspensions and incubating them at 25°C for 7 days. Suspension of conidia was prepared by suspending mycelia scraped from 7-day-old cultures of pathogenic fungi in PDA. The resulting suspensions were filtered through a double layer of cheesecloth. The concentration of spore suspensions was adjusted to 105 conidia/ml using a hemocytometer before application.
The selected P. polymyxa strain APEC128 was subjected to molecular identification using sequence homology of its 16S rRNA gene (Weisburg et al., 1991). Genomic DNA of APEC128 was isolated using a Genomic DNA Extraction Kit for bacteria (iNtRON Biotechnology, Seongnam, Korea) following manufacturer’s instructions. The 16S rRNA gene was amplified by polymerase chain reaction (PCR) with Taq DNA polymerase and primers 27F (5′-AGAGTTTGATCMTGGCTCAG-3′) and 1492R (5′-GGYTACCTTGTTACGACTT-3′). The conditions for thermal cycling were as follows: denaturation at 94°C for 5 minutes followed by 30 cycles at 94°C for 1 minute, annealing at 56°C for 1 minute and extension at 72°C for 1 minute. At the end of the cycling, the reaction mixture was held at 72°C for 5 minutes and then cooled to 4°C. The obtained PCR products were sequenced with an automated sequencer (Genetic Analyzer 3130; Applied Biosystems, Foster City, CA, USA) using the same primers. The sequences were compared for similarity with the reference species of bacteria available in genomic database using the National Center for Biotechnology Information (NCBI)-BLAST tool. Sequence alignment and phylogenic tree construction were performed using a MEGA 4.0 program (Tamura et al., 2007).
To determine the growth of the strain APEC128 in different growth media, a single colony of APEC128 was inoculated into a test tube (20 mm diameter) containing 5 ml of growth medium and incubated for 24 hours at 28°C under shaking condition (250 rpm). The broth (5 ml) was transferred into a 500-ml baffled flask containing 100 ml growth medium and incubated at 28°C for 48 hours with shaking at 250 rpm. The various tested media included BHI media, potato dextrose broth, NB, tryptic soy broth (TSB), and Luria-Bertani broth. Samples were collected at 4-hour intervals. Cell growth was expressed as a measure of turbidity at 600 nm using a spectrophotometer (Ultrospec 4000 Spectrophotometer; Pharmacia Biotech Ltd., Little Chalfont, UK). For optimization of pH and temperature conditions, the isolate APEC128 was cultured at different incubation temperatures (15°C, 20°C, 25°C, 30°C, 35°C, 40°C, and 45°C). All cultures were incubated on a rotary shaker at 180 rpm at different temperatures for 48 hours and absorbance was read at 600 nm. For optimization of pH, the broth was checked for different pH (3, 5, 7, and 9) before autoclaving. The culture broths were inoculated with the bacterial suspensions (108 cfu/ml), and the cultures were incubated under shaking condition (180 rpm) at 28°C for 48 hours and the absorbance was read at 600 nm.
Chitinase assay was conducted according to the method developed by Roberts and Selitrennikoff (1988). In brief, the bacterial culture was spotted on prepared minimal agar plates amended with 0.3% colloidal chitin and incubated at 30°C for 7 days. Development of a halo zone around the colony after addition of iodine was considered positive for chitinase enzyme activity. Amylolytic activity assay was performed according to the previously described method (Shaw et al., 1995). Twenty-four-hour-old bacterial culture spot inoculated on starch agar plate and incubated at 37°C for 24 to 48 hours. The petridishes were flooded with iodine solution and observed for starch hydrolysis, which was indicated by the formation of a clear zone (diameter, mm) around the bacterial spot. The cellulase activity assay was carried out qualitatively using carboxymethylcellulose (CMC) as the sole carbon and energy source (Hankin and Anagnostakis, 1977). CMC agar plates were inoculated with 24-hour-old grown bacterial cultures and incubated at 28°C for 48 hours. The halo zone due to cellulose hydrolysis around the colony was visualized by staining with 1% Congo red dye for 15 minutes, followed by destaining with 1 M NaCl solution for 20 minutes. The clear halo zone around the bacterial colony indicated the CMC hydrolyzing capacity. Proteolytic assay was performed according to the method described by Fleming et al. (1975). Briefly, proteolytic activity of APEC128 was determined by spot inoculation of bacterial culture (72-hour-old) on skim milk agar (100 ml of NA supplemented with sterilized skim milk) and incubation at 28°C for 24 to 48 hours. Formation of a clear zone (diameter, mm) around the bacterial spot was considered a positive test for proteolysis. Each treatment consisted of five replicates, and each experiment was repeated at least two times.
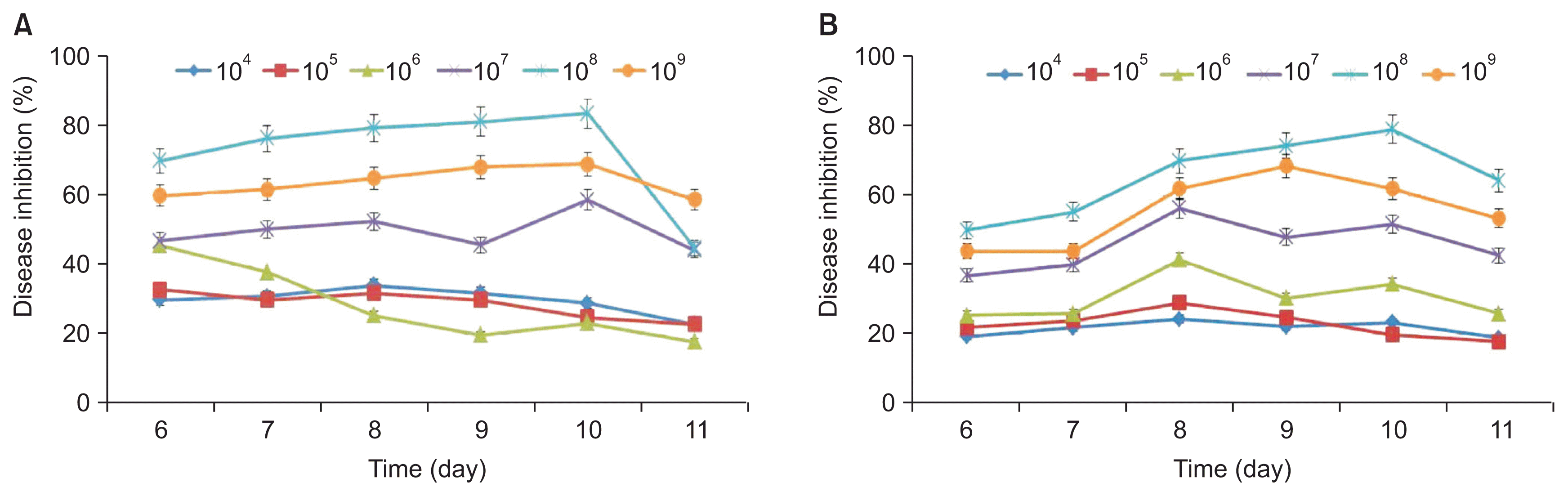
Apple fruits cv. ‘Fuji’ of similar size were selected for our study. The fruits were surface-sterilized with 70% ethanol followed by 2% NaOCl for 5 minutes, and then washed three times with SDW and air-dried in a laminar air flow chamber. The effect of bacterial inoculum density on the development of anthracnose disease was tested by inoculating apple fruits with various concentrations of APEC128 suspensions (104, 105, 106, 107, 108, and 109 cfu/ml). Surface-sterilized apple fruits were wounded by piercing them 1 to 2 mm deep with a sterile pin inoculated with 10 μl of inoculum and allowed to dry for 10 minutes. The spore suspensions (10 μl) of the pathogenic fungi were inoculated onto the wounds of the apple fruits. The diameter of the symptoms was observed 6 to 11 days after incubation at 25°C and compared with the non-treated control (without bacterial suspension). Each treatment consisted of 18 replicates (fruits), and each experiment was repeated three times.
The data were subjected to ANOVA using SAS JMP software ver. 3 (SAS Institute, Cary, NC, USA; SAS Institute, 1995). Significant differences between the treatment means were determined using the least significant difference at P < 0.05. All the experiments were performed at least two times. For each experiment, the data were analyzed separately, and the results of one representative experiment are shown.
The isolate APEC128 was selected based on the primary screening of antagonistic activity against C. gloeosporioides under in vitro conditions using a disc diffusion assay (Fig. 1). APEC128 was further characterized by 16S rRNA gene sequencing. Comparison of the specific sequence of the ribosomal gene with sequences deposited in GenBank suggested that the APEC128 isolate belonged to the Paenibacillus genus with the highest homology to P. polymyxa (99.8%). In the phylogenetic tree, APEC128 was clustered with other Paenibacillus spp., and it was sister to t P. polymyxa (bootstrap support of 42%) (Fig. 2). The comparative sequence analysis of 16S rRNA gene (1,480 bp) of APEC128 revealed that it was the member of the genus Paenibacillus.
The antagonistic activity of APEC128 against C. gloeosporioides was assessed using the disc diffusion assay. Growth inhibition of phytopathogenic mycelia in culture plates was observed 5 days after inoculation. The strain producing the largest zone of fungal growth inhibition (18.5 mm) during the screening under in vitro conditions was identified as P. polymyxa APEC128. The antagonist strongly inhibited the development of anthracnose caused by C. gloeosporioides and C. acutatum (Fig. 3) compared to other strains tested, suggesting that strain APEC128 could significantly inhibit the growth of the two fungal pathogens.
The APEC128 cell numbers were relatively higher when cultured in BHI broth as compared to other media 48 hours after incubating at 28°C (Fig. 4). The next optimum growth medium was TSB. The smallest number of bacterial cells of APEC128 was observed when the cells were cultured in NB medium. The results clearly showed that BHI was the optimal medium to support the most efficient proliferation of APEC128 cells. The optimal pH and temperature conditions for growth of APEC128 were determined for the BHI medium. Thus a greater bacterial cell growth was observed at pH 8 (Fig. 5A) and a temperature of 30°C (Fig. 5B). The optimum temperature was 30°C for better bacterial cell growth in comparison with other temperature conditions (Fig. 5B). Interestingly, bacterial growth at 25°C was identical to that at 35°C, but it was drastically diminished at temperatures above 35°C, as measured by turbidity after incubation.
The strain APEC128 was evaluated for its potential to produce various enzymes in vitro. APEC128 produced amylase and protease at a greater level, compared to chitinase and cellulase, which were secreted only at a minor level, as indicated by the zone of hydrolysis around the bacterial colonies (Fig. 6). These results suggested that the strain APEC128 had a potential for production of various enzymes in vitro, which contribute to various biological activities.
Optimal inoculum concentration suppressed the disease development at a maximum level. Fig. 7 shows that in apples treated with APEC128 cells at the concentration of 1 × 108 cfu/ ml, anthracnose caused by C. gloeosporioides and C. acutatum was suppressed by 83.6% (Fig. 7A) and 79% (Fig. 7B), respectively, 10 days after inoculation. Apples treated with strain APEC128 at the concentration of either 1 × 106 or 1 × 107 cfu/ml showed rot symptoms, and the disease was suppressed by < 50% on the 6th day after inoculation. These results confirmed the biocontrol activity of the strain APEC128 against C. gloeosporioides and C. acutatum in vivo, which activity was positively correlated with the concentrations used. To investigate the potential for biological control activity of APEC128 against these two pathogens in vivo; the apples were treated with a bacterial suspension prior to fungal pathogen exposure.
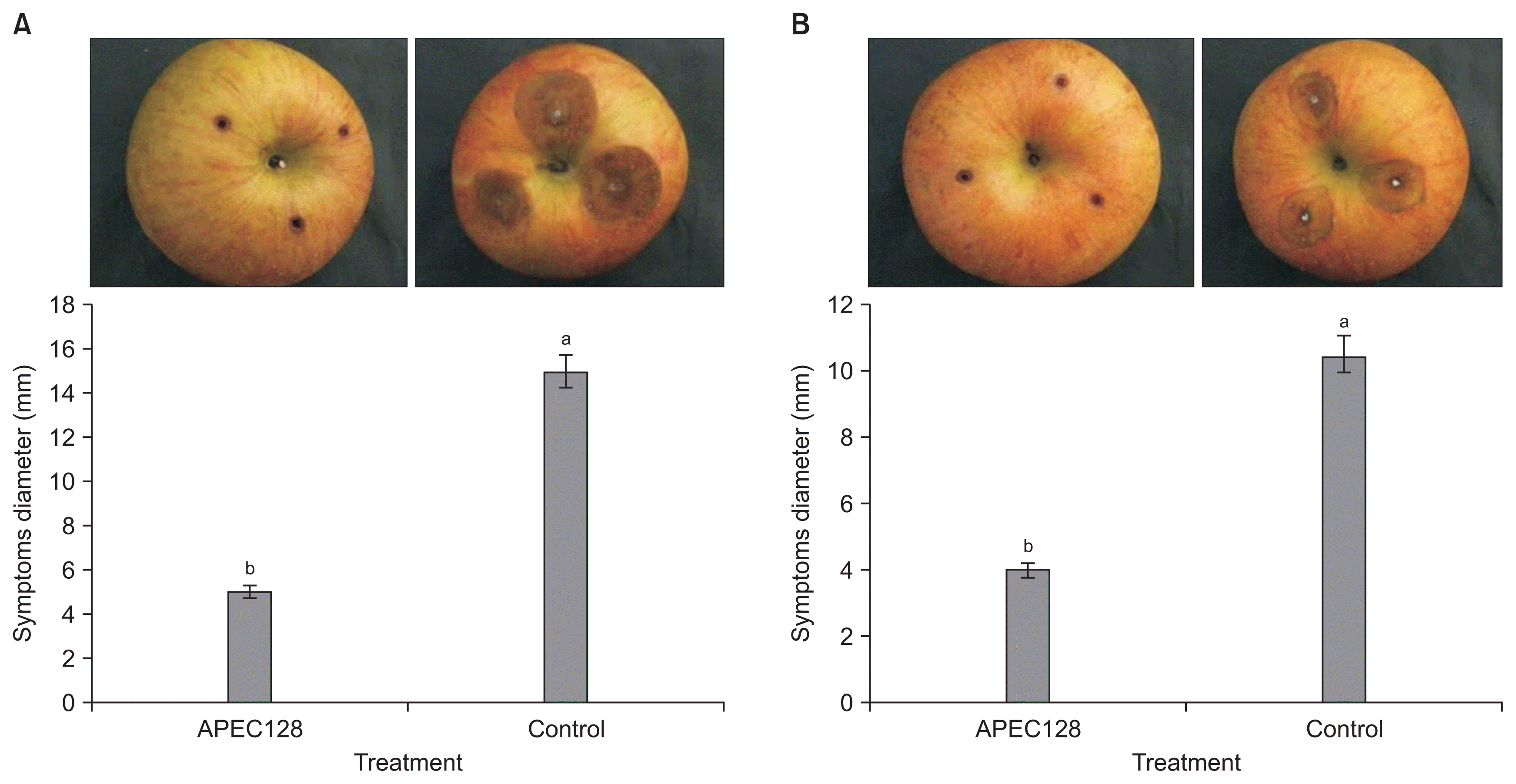
The symptoms started to appear 7 days post-inoculation when apples were soaked in a bacterial suspension at a concentration of 1 × 108 cfu/ml prior to inoculation with spore suspensions. There was a greater suppression of disease development of C. gloeosporioides and C. acutatum in APEC128-treated apple fruits when compared to non-treated control. The lesion areas caused by C. gloeosporioides and C. acutatum were 5.2 mm (Fig. 8A) and 4.4 mm (Fig. 8B) in diameter, respectively, where those non-treated apples were about 3-fold greater compared to lesions in APEC128-treated apples.
Biological control of various plant diseases using microorganisms is an effective approach to control diseases in an eco-friendly manner. The first step of this process is to screen for potential biological control agents, and the main screening method under in vitro conditions is based on antagonistic activity. Spores are the major inoculants both in the field and in the post-harvest period for many plant diseases, including apple anthracnose. Our results confirmed a strong inhibitory activity of bacterial cells of APEC128 on the mycelial growth of fungal pathogens C. gloeosporioides and C. acutatum (Fig. 3). These results are consistent with previous reports (Lee et al., 2012; Li et al., 2009) and indicated that spore germination of several pathogens is inhibited under in vitro condition. At present, the use of post-harvest biocontrol agents is constrained by the lack of consistent efficacy and the high level of control required during post-harvest conditions. Recently, there has been an increased interest in further development of commercial products. The strain APEC128 isolated in this study protected apples from the post-harvest disease anthracnose at the required concentration of antagonistic bacterial cells.
Saravanakumar et al. (2008) reported that yeast Metschnikowia pulcherrima is an effective biocontrol agent that inhibited several fungal pathogens on harvested apples in low temperatures under controlled conditions. The yeast out-competes fungal pathogens for iron available in the rhizosphere soil, thus restricting fungal growth since iron is essential for their fungal growth and pathogenesis. Previously, the microorganism Bacillus subtilis has been reported to control diseases in harvested fruits (Yánez-Mendizábal et al., 2011). Its biocontrol activity is mainly due to the production of antifungal compounds such as antibiotics, surfactin, iturin and gramicidin (Cho et al., 2003). In addition, a previous study by Sadhana and Silvia (2009) has demonstrated that P. polymyxa strain P13 isolated from fermented sauce secretes a compound referred to polyxin exhibited the antagonistic activity against Lactobacillus strains; and Xu et al. (2014) showed that Paenibacillus kribbensis exhibited antagonistic activity against fungal pathogens by secreting lytic enzymes, which has the ability to break down fungal cell walls. Similarly, another report by Kim et al. (2009) stated that Paenibacillus spp., produce a variety of secondary metabolites such as antibiotics, siderophores, hydrogen cyanide, and a variety of enzymes. Herein, we show that pH and temperature were important factors for the optimum growth of the strain APEC128. On the other hand, chitinase produced by various microbial source is involved in biological control of fungal diseases since it degrades chitin, a major component of the fungal cell wall, thus inactivating pathogenic fungi (Agrawal and Kotasthane, 2012). Furthermore, production of other enzymes by bacteria, such as amylase (Fossi et al., 2009), protease and cellulase by Paenibacillus spp. may be involved in suppressing diseases caused by pathogenic fungi (Alvarez et al., 2006; Liang et al., 2014), and these results are further supported by a recent study (Han et al., 2015), stated that protease and cellulase production by B. amyloliquefaciens responsible for the antagonistic activity.
Multiple modes of action of Paenibacillus spp. were assumed to contribute to the successful biocontrol activity, including competition for nutrients, induced resistance, and production of antibiotics and cell wall-degrading enzymes such as chitinases and β-1,3-glucanase (Droby et al., 2009). The results presented herein indicate similar modes of action by the strain APEC128 against anthracnose- causing pathogens. Many studies have reported the different antifungal compounds produced by Bacillus spp. and their importance in biocontrol activity (Lee et al., 2009; Liu et al., 2011). The strain APEC128 strongly inhibited the growth of fungal pathogens in vitro, probably due to its production of antifungal compounds. Previously, Janisiewicz and Korsten (2002) reported that the lytic enzymes produced by Bacillus spp. are involved in the biological control activity.
In conclusion, this study demonstrates that APEC128 is an effective biocontrol agent against apple anthracnose in vitro and in vivo. Moreover, the post-harvest treatment of apples with a bacterial suspension of APEC128 significantly suppressed the disease development during the storage period. Our results indicate that APEC128 is one of the promising agents for the biocontrol of apple anthracnose both in the field and after harvest. Further investigation will elucidate its mechanism of biocontrol ability on apple fruits.
Acknowledgments
This work was supported by a grant from 2011 Research Fund of Andong National University, Korea.
Fig. 1
In vitro screening of antagonistic activity of 30 bacterial isolates, including APEC128 against mycelial growth of anthracnose caused by Colletotrichum gloeosporioides by disc diffusion assay. The inhibition zone was measured 5 days after incubating at 25°C. The experiment was conducted at least twice with eight replications per treatment with similar results. Bars indicate the standard error of the mean, and bars with the same letters do not differ significantly between each other according to the least significant difference (P < 0.05).
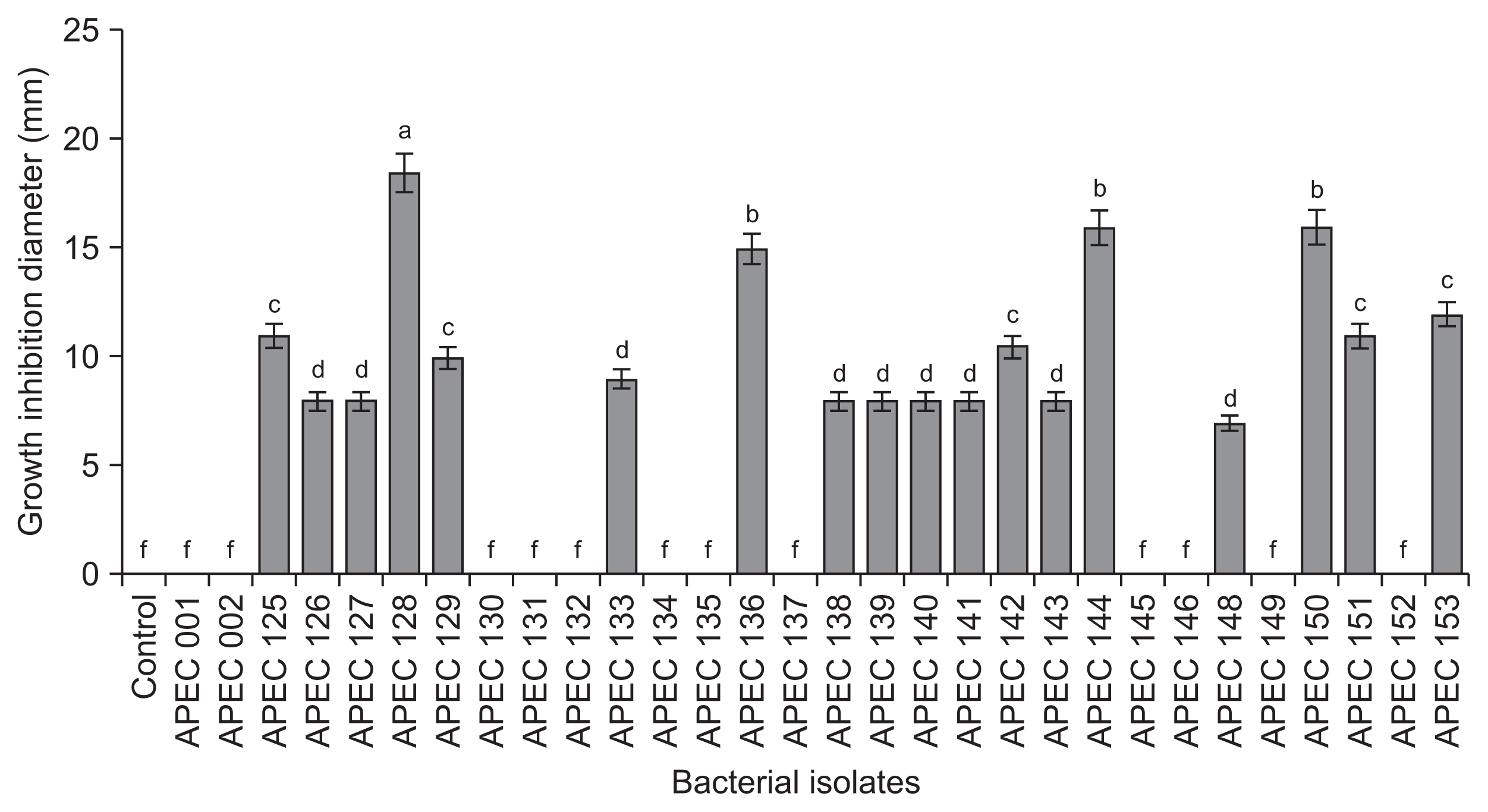
Fig. 2
Phylogenetic dendrogram inferred from the 16S rRNA gene sequences showing the relationships between Paenibacillus polymyxa strain APEC128 (APEC128) and related Paenibacillus species. Bootstrap values (expressed as percentages of 1,000 replications) greater than 50% are shown at branch points and the species names are preceded by the National Center for Biotechnology Information GenBank accession numbers. Phylogenetic tree was generated using maximum parsimony implemented in MEGA 4.0 program. The scale bar indicates 0.02 substitutions per nucleotide position.
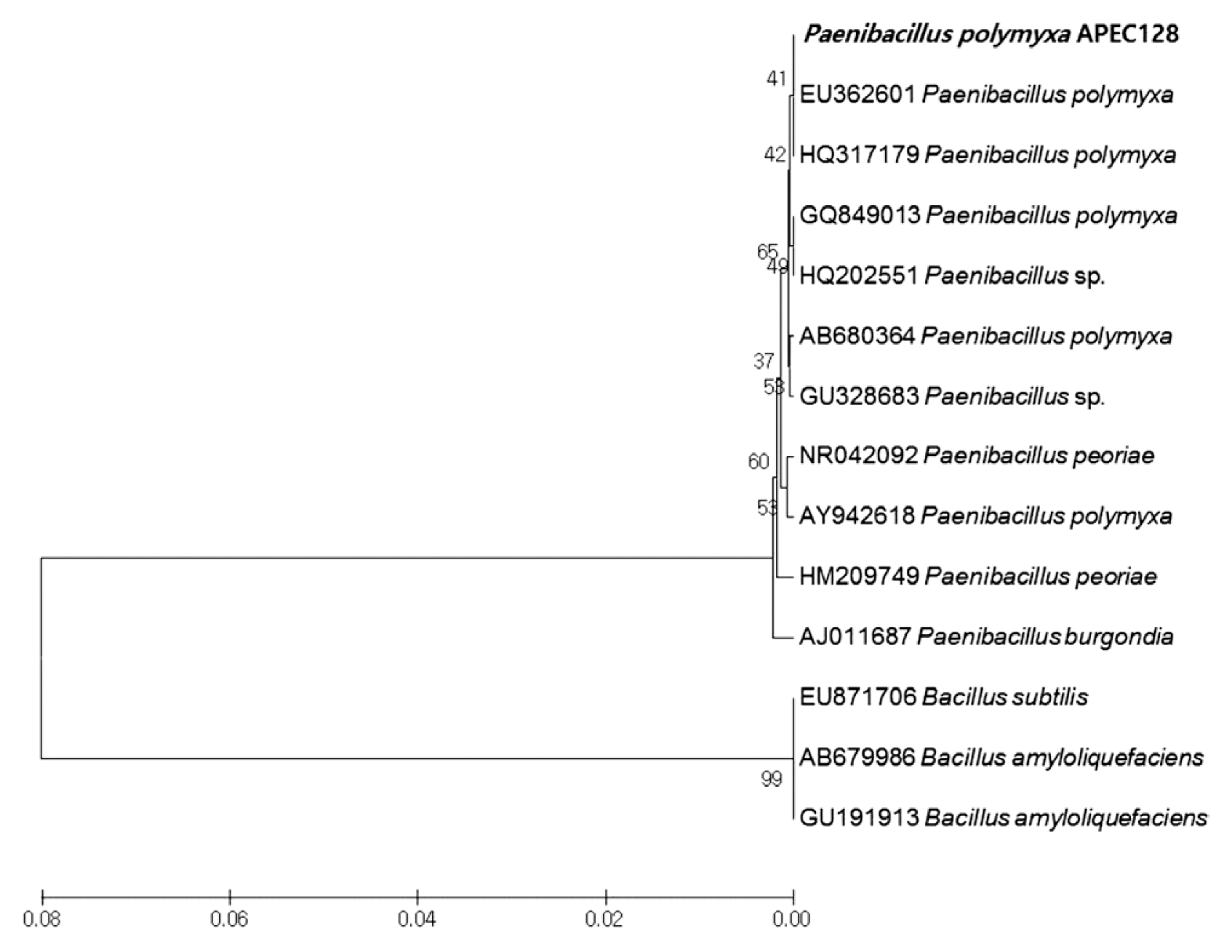
Fig. 3
In vitro antagonistic activity of Paenibacillus polymyxa strain APEC128 (asterisks) against phytopathogens, Colletotrichum gloeosporioides and C. acutatum, assessed with the disc diffusion assay. Photographs were taken after 5 days of incubation at 25°C. The experiment was repeated at least two times.
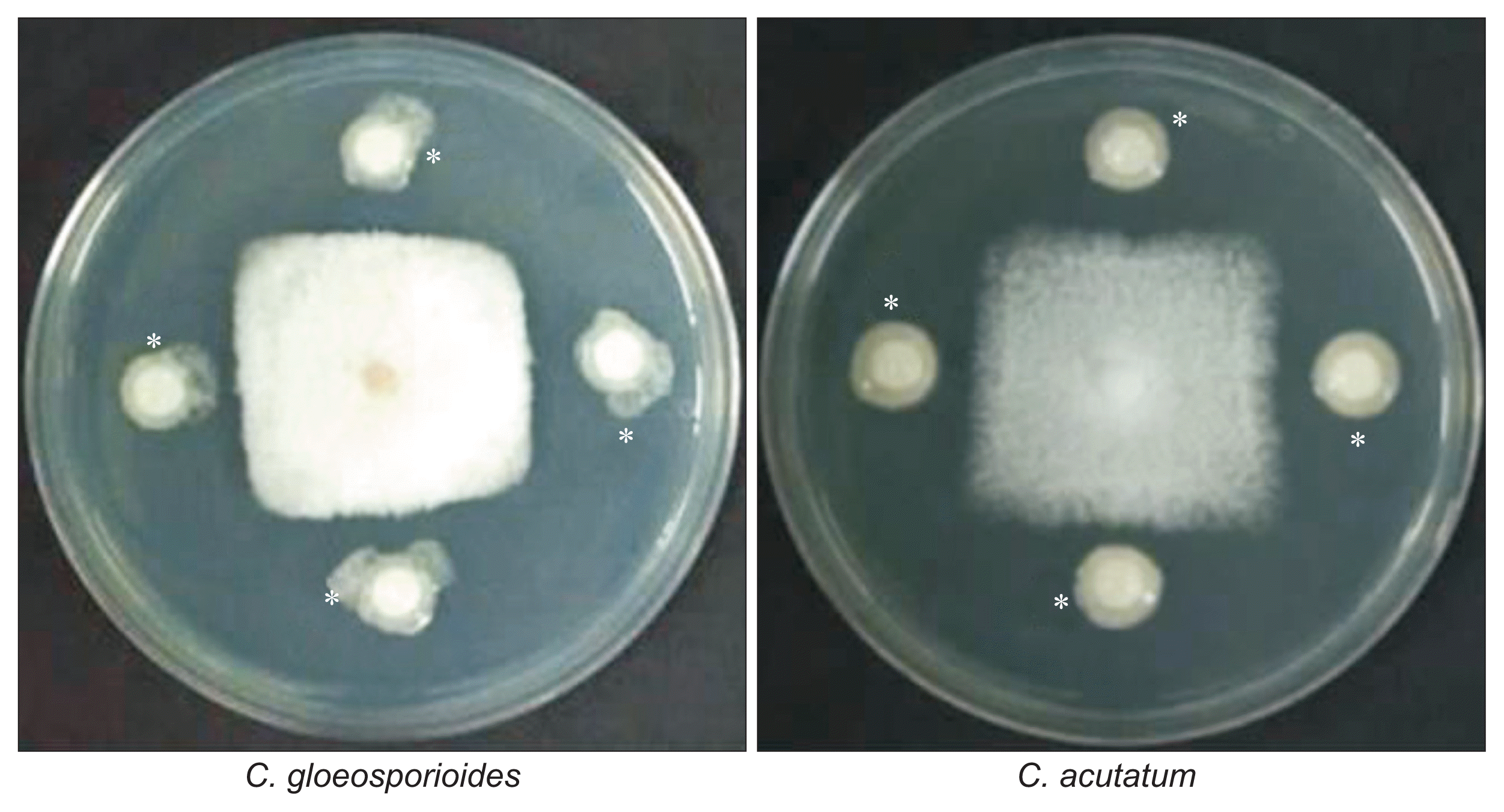
Fig. 4
Effect of various growth media on APEC128. Optical density (OD) was read at 600 nm 48 hours after incubation at 28°C. The experiment was repeated three times. Bars with the same letters do not differ significantly according to the least significant difference (P < 0.05). TSB, tryptic soy broth; BHI, brain-heart infusion; LB, Luria-Bertani broth; PDB, potato dextrose broth; NB, nutrient broth.
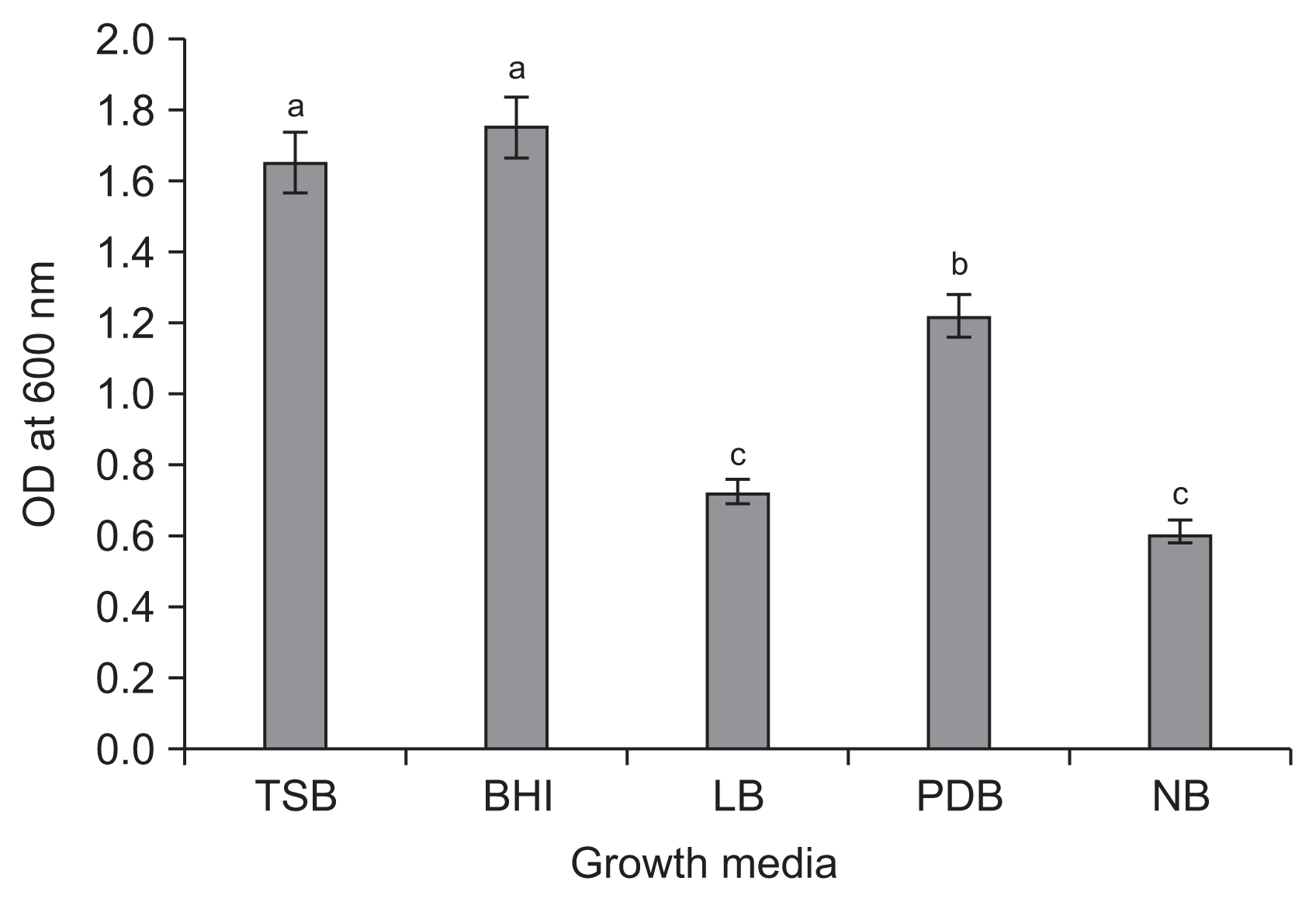
Fig. 5
Effect of pH (A) and temperature (B) on the growth of APEC128 cells. The experiment was repeated at least two times. Values are mean ± standard error.
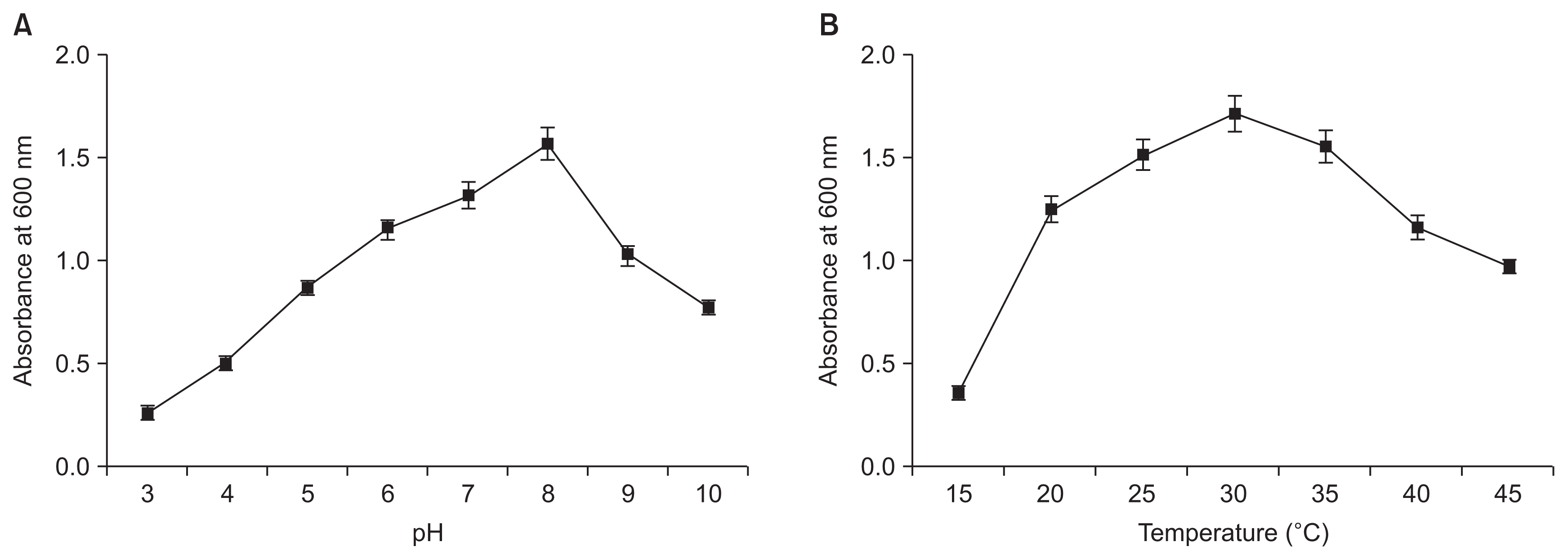
Fig. 6
The level of chitinase, amylase, cellulase, and protease activity in vitro by APEC128 inferred from the size of the halo zones. The experiment was repeated at two times. Bars with the same letters do not differ significantly according to the least significant difference (P < 0.05).

References
Afanador-Kafuri, L, Minz, D, Maymon, M and Freeman, S 2003. Characterization of Colletotrichum isolates from tamarillo, passiflora, and mango in Colombia and identification of a unique species from the genus. Phytopathology. 93:579-587.


Agrawal, T and Kotasthane, AS 2012. Chitinolytic assay of indigenous Trichoderma isolates collected from different geographical locations of Chhattisgarh in Central India. SpringerPlus. 1:73




Alvarez, VM, von der Weid, I, Seldin, L and Santos, AL 2006. Influence of growth conditions on the production of extracellular proteolytic enzymes in Paenibacillus peoriae NRRL BD-62 and Paenibacillus polymyxa SCE2. Lett Appl Microbiol. 43:625-630.


Ash, C, Priest, FG and Collins, MD 1993. Molecular identification of rRNA group 3 bacilli (Ash, Farrow, Wallbanks and Collins) using a PCR probe test. Proposal for the creation of a new genus Paenibacillus. Antonie van Leeuwenhoek. 64:253-260.



Bajpai, VK, Choi, SW, Cho, MS and Kang, SC 2009. Isolation and morphological identification of apple anthracnose fungus of Colletotrichum sp. KV-21. Korean J Environ Agri. 28:442-446.

Cho, SJ, Lee, SK, Cha, BJ, Kim, YH and Shin, KS 2003. Detection and characterization of the Gloeosporium gloeosporioides growth inhibitory compound iturin A from Bacillus subtilis strain KS03. FEMS Microbiol Lett. 223:47-51.


Cook, RJ 2000. Advances in plant health management in the twentieth century. Annu Rev Phytopathol. 38:95-116.


Deacon, JW 1997. Modern mycology. Blackwell Scientific, Oxford, UK.
Dijksterhuis, J, Sanders, M, Gorris, LG and Smid, EJ 1999. Antibiosis plays a role in the context of direct interaction during antagonism of Paenibacillus polymyxa towards Fusarium oxysporum. J Appl Microbiol. 86:13-21.


Droby, S, Wisniewski, M, Macarisin, D and Wilson, C 2009. Twenty years of postharvest biocontrol research: is it time for a new paradigm? Postharvest Biol Technol. 52:137-145.

Ferreira, JHS, Matthee, FN and Thomas, AC 1991. Biological control of Eutypa lata on grapevine by an antagonistic strain of Bacillus subtilis. Phytopathology. 81:283-287.

Fleming, HP, Etchells, JL and Costilow, RN 1975. Microbial inhibition by an isolate of Pediococcus from cucumber brines. Appl Microbiol. 30:1040-1042.




Fossi, BT, Tavea, F, Jiwoua, C and Ndjouenkeu, R 2009. Screening and phenotypic characterization of thermostable amylases producing yeasts and bacteria strains from some Cameroonian soils. Afr J Microbiol Res. 3:504-514.
Gouzou, L, Burtin, G, Philippy, R, Bartoli, F and Heulin, T 1993. Effect of inoculation with Bacillus polymyxa on soil aggregation in the wheat rhizosphere: preliminary examination. Geoderma. 56:479-491.

Han, JH, Shim, H, Shin, JH and Kim, KS 2015. Antagonistic activities of Bacillus spp. strains isolated from tidal flat sediment towards anthracnose pathogens Colletotrichum acutatum and C. gloeosporioides in South Korea. Plant Pathol J. 31:165-175.



Hankin, L and Anagnostakis, SL 1977. Solid media containing carboxymethylcellulose to detect CX cellulose activity of micro-organisms. J Gen Microbiol. 98:109-115.

Henz, GP, Boiteux, LS and Lopes, CA 1992. Outbreak of strawberry anthracnose caused by Colletotrichum acutatum in central Brazil. Plant Dis. 76:212

Heulin, T, Berge, O, Mavingui, P, Gouzou, L, Hebbar, KP and Balandrean, J 1994. Bacillus polymyxa and Rahnella aquatilis, the dominant N2-fixing bacteria associated with rhizosphere in French soils. Eur J Soil Biol. 30:35-42.
Janisiewicz, WJ and Korsten, L 2002. Biological control of postharvest diseases of fruits. Annu Rev Phytopathol. 40:411-441.


Kim, SG, Khan, Z, Jeon, YH and Kim, YH 2009. Inhibitory effects of Paenibacillus polymyxa GBR-462 on Phytophthora capsici causing phytophthora blight in chili pepper. J Phytopathol. 153:329-337.
Kloepper, JW 1992. Plant growth-promoting rhizobacteria as biological control agents. In: Soil microbial ecology: applications in agricultural and environmental management, eds. by FB Metting, 225-274. Marcel Dekker Inc, New York, NY, USA.
Lee, DH, Kim, DK, Jeon, YA, Uhm, JY and Hong, SB 2007. Molecular and cultural characterization of Colletotrichum spp. causing bitter rot of apples in Korea. Plant Pathol J. 23:37-44.

Lee, GW, Ko, JA, Oh, BT, Choi, JR, Lee, KJ, Chae, JC and Kamala-Kannan, S 2012. Biological control of postharvest diseases of apples, peaches and nectarines by Bacillus subtilis S16 isolated from halophytes rhizosphere. Biocontrol Sci Technol. 22:351-361.

Lee, KY, Heo, KR, Choi, KH, Kong, HG, Nam, J, Yi, YB, Park, SH, Lee, SW and Moon, BJ 2009. Characterization of a chitinase gene exhibiting antifungal activity from a biocontrol bacterium Bacillus licheniformis N1. Plant Pathol J. 25:344-351.

Li, J, Yang, Q, Zhao, LH, Zhang, SM, Wang, YX and Zhao, XY 2009. Purification and characterization of a novel antifungal protein from Bacillus subtilis strain B29. J Zhejiang Univ Sci B. 10:264-272.




Liang, YL, Zhang, Z, Wu, M, Wu, Y and Feng, JX 2014 Isolation, screening, and identification of cellulolytic bacteria from natural reserves in the subtropical region of China and optimization of cellulase production by Paenibacillus terrae ME27-1. Biomed Res Int. Online publication.


Liu, J, Zhou, T, He, D, Li, XZ, Wu, H, Liu, W and Gao, X 2011. Functions of lipopeptides bacillomycin D and fengycin in antagonism of Bacillus amyloliquefaciens C06 towards Monilinia fructicola. J Mol Microbiol Biotechnol. 20:43-52.

Mari, M, Neri, F and Bertolini, P 2009. Management of important diseases in Mediterranean high value crops. Stewart Postharvest Rev. 5:1-10.

Mavingui, P and Heulin, T 1994. In vitro chitinase and antifungal activity of a soil, rhizosphere and rhizoplane population of Bacillus polymyxa. Soil Biol Biochem. 26:801-803.

Nunes, C, Usall, J, Teixido, N, Torres, R and Vinas, I 2002. Control of Penicillium expansum and Botrytis cinerea on apples and pears with the combination of Candida sake and Pantoea agglomerans. J Food Prot. 65:178-184.



Onofre, SB and Antoniazzi, D 2014. Behavior of the fungus Colletotrichum gloeosporioides (Penz & Sacc.), which causes bitter rot in apples after harvesting. Adv Microbiol. 4:202-206.
Park, JW, Balaraju, K, Kim, JW, Lee, SW and Park, KS 2013. Systemic resistance and growth promotion of chili pepper induced by an antibiotic producing Bacillus vallismortis strain BS07. Biol Control. 65:246-257.

Roberts, WK and Selitrennikoff, CP 1988. Plant and bacterial chitinases differ in antifungal activity. J Gen Microbiol. 134:169-176.

Rosado, AS and Seldin, L 1993. Production of a potentially novel anti-microbial substance by Bacillus polymyxa. World J Microbiol Biotechnol. 9:521-528.



Sadhana, L and Silvia, T 2009. Ecology and biotechnological potential of Paenibacillus polymyxa: a minireview. Indian J Microbiol. 49:2-10.




Saravanakumar, D, Ciavorella, A, Spadaro, D, Garibaldi, A and Gullino, ML 2008. Metschnikowia pulcherrima strain MACH1 outcompetes Botrytis cinerea, Alternaria alternata and Penicillium expansum in apples through iron depletion. Postharvest Biol Technol. 49:121-128.

SAS Institute. 1995. JMP statistics and graphics guide, version 3. SAS Institute, Cary, NC, USA. 65-95.
Shaw, J, Lin, FP, Chen, SC and Chen, HC 1995. Purification and properties of an extracellular α-amylase from Thermus sp. Bot Bull Acad Sin. 36:195-200.
Shishido, M, Massicotte, HB and Chanway, CP 1996. Effect of plant growth promoting Bacillus strains on pine and spruce seedling growth and mycorrhizal infection. Ann Bot. 77:433-442.

Singh, HP and Singh, TA 1993. The interaction of rockphosphate Bradyrhizobium, vesicular-arbuscular mycorrhizae and phosphate-solubilizing microbes on soybean grown in asub-Himalayan mollisol. Mycorrhiza. 4:37-43.


Tamura, K, Dudley, J, Nei, M and Kumar, S 2007. MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol. 24:1596-1599.


Víchová, J, Staňková, B and Pokorný, R 2012. First report of Colletotrichum acutatum on tomato and apple fruits in the Czech Republic. Plant Dis. 96:769-770.

Weisburg, WG, Barns, SM, Pelletier, DA and Lane, DJ 1991. 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol. 173:697-703.




- TOOLS
-
METRICS

- Related articles
-
Biological Control of Rice Bakanae by an Endophytic
Bacillus oryzicola YC70072016 June;32(3)Biological Control of
Meloidogyne hapla Using an Antagonistic Bacterium2014 September;30(3)Biological Control of Apple Ring Rot on Fruit by
Bacillus amyloliquefaciens 90012013 June;29(2)



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



