 |
 |
| Plant Pathol J > Volume 34(6); 2018 > Article |
Abstract
Clubroot is one of the most economically important diseases of the Brassicaceae family. Clubroot disease is caused by the obligate parasite Plasmodiophora brassicae, which is difficult to study because it is non-culturable in the laboratory and its races are genetically variable worldwide. In Korea, there are at least five races that belongs to four pathotype groups. A recent study conducted in Korea attempted to develop molecular markers based on ribosomal DNA polymorphism to detect P. brassicae isolates, but none of those markers was either race-specific or pathotype-specific. Our current study aimed to develop race- and isolate-specific markers by exploiting genomic sequence variations. A total of 119 markers were developed based on unique variation exists in genomic sequences of each of the races. Only 12 markers were able to detect P. brassicae strains of each isolate or race. Ycheon14 markers was specific to isolates of race 2, Yeoncheon and Hoengseong. Ycheon9 and Ycheon10 markers were specific to Yeoncheon isolate (race 2, pathotype 3), ZJ1-3, ZJ1-4 and ZJ1-5 markers were specific to Haenam2 (race 4) isolate, ZJ1-35, ZJ1-40, ZJ1-41 and ZJ1-49 markers were specific to Hoengseong isolate and ZJ1-56 and ZJ1-64 markers were specific to Pyeongchang isolate (race 4, pathotype 3). The PCR-based sequence characterized amplified region (SCAR) markers developed in this study are able to detect five Korean isolates of P. brassicae. These markers can be utilized in identifying four Korean P. brassicae isolates from different regions. Additional effort is required to develop race- and isolate-specific markers for the remaining Korean isolates.
Clubroot is a serious disease of the Brassicaceae that is increasingly problematic worldwide (Wang et al., 2008). This disease is caused by the soil-borne biotrophic cercozoan Plasmodiophora brassicae Woronin (Nikolaev et al., 2004). This pathogen lives in soil for up to 20 years as resting spores (Karling, 1968; Wallenhammar, 1996) that are difficult to control with anti-microbial chemicals or crop rotation (Tsushima, 2000; Voorrips, 1995). Root hairs are the primary target of this pathogen, but the infection eventually spreads to the cortical region of the roots. The infected roots swell to form of galls or clubs that severely reduce the quality and production of Brassica crops, such as cabbage (Dixon, 2009; Kim et al., 2009). Several factors increase the likelihood of P. brassicae infection, including acidification of soils, changes in climate that favor the growth of this pathogen, an increase in cultivated area devoted to Brassica crops, and transportation of cruciferous crop products and residues (Li et al., 2013). The resting spores multiply exponentially in the newly infected soil if they parasitize a cruciferous crop.
Understanding of clubroot disease has been limited by several aspects of the pathogen’s biology. P. brassicae is non-culturable in laboratory conditions and is difficult to clone and sequence due to its high genomic similarity with host plants (Bryngelsson et al., 1988; Laila et al., 2017; Schwelm et al., 2015). Moreover, several pathotypes (a group of organisms that have the same pathogenicity on a specified host) and races of P. brassicae exist worldwide (Buczacki et al., 1975; Hatakeyama et al., 2004; Kuginuki et al., 1999; Somé et al., 1996; Williams, 1966). Kim et al. (2016) re-classified Korean P. brassicae isolates into four pathotypes (Jo et al., 2011; Kim et al., 2016). Development of race-specific markers would enable more effective and precise detection of this pathogen. Such molecular markers have been used effectively in plant disease diagnostics for many years, including limited methods in P. brassicae based on PCR amplification of DNA from galls or resting spores (Faggian and Strelkov, 2009; Ito et al., 1997, 2008; Li et al., 2017; Wallenhammar and Arwidsson, 2001; Yang et al., 2002). Ito et al. (1997) cloned and sequenced 1.4 kb DNA fragment from 19 different Japanese P. brassicae isolates and found that the markers covering that sequence do not amplify fungi and bacterial DNA they studied. Thus, the authors suggested two markers PBTZS-3 and PBTZS-4 for detecting 19 P. brassicae isolates. This research group also developed a single-tube nested polymerase chain reaction method for detecting P. brassicae isolates from soil samples. In another study conducted in Sweden, Wallenhammar and Arwidsson (2001) found two markers of 389 bp and 507 bp were able to detect P. brassicae isolates from soil sample of both Sweden and UK by a nested PCR method. The authors, however, found that isolates from two different countries had variation in 18s and ITS1 (inter-transcribed spacer 1) regions indicating that those markers are unable to differentiate any isolate specific variations. In a recent study in China, Li et al. (2017) successfully detected an unknown P. brassicae isolate after targeting D85819 and Pro1 gene-specific primers. However, none of those PCR based methods could distinguish between the different P. brassicae isolates. P. brassicae isolates in Korea differ in their level of pathogenicity and virulence (Laila, 2018). It might be also possible that two or more isolates of from adjacent geographical regions might co-exist in a particular location. Furthermore, isolates from one region may translocate to another region due to transportations of green plant products. Therefore, an effective detection protocol of existing Korean isolates is important for their race-specific and isolate-specific identity. Laila et al. (2017) attempted to distinguish 11 Korean P. brassicae isolates by creating ribosomal DNA (rDNA) markers. This approach produced four high resolution melting-based markers targeting smaller subunit region of ribosomal DNA that can detect two groups of the 11 P. brassicae isolates. However, none of these markers could distinguish between the Korean isolates. In this study, we compared all available genomic sequences of P. brassicae to obtain isolate-specific molecular markers.
Genomic sequences of P. brassicae isolates e3 and ZJ-1 were retrieved from NCBI (Genbank accession numbers GCA_001049375.1 and GCA_002093825.1, respectively). The sizes of the e3 and ZJ-1 genomes were 24.0508 and 24.8311 Mb, respectively (Table 1). We recently sequenced partial genomic DNA sequences of four additional P. brassicae isolates (21 Mb each): Daejeon, Gangneung 1, Haenam-2, and Yeoncheon (SRA accession: SRP150814). Genomic sequences of these four isolates were compared to identify variable nucleotide positions. These six sequences were aligned using the “Align with progressiveMauve” approach in Mauve (v. 2.4.0) (Darling et al., 2010) and visualized in Geneious (v. 11.0.3, Kearse et al., 2012). This comparison identified variable regions among these isolates that were used to develop isolate-specific primers (Fig. 1).
Isolate-specific primers were designed using Primer3 software (Koressaar and Remm, 2007; Untergasser et al., 2012). Primer specificity was tested following Robin et al. (2016). A total of 115 primers were designed (Table 2, Supplementary Table 1).
We infected Chinese cabbage variety Bullam-3-ho with 11 previously collected P. brassicae isolates: Gangneung (1 and 2), Yeoncheon, Daejeon, Haenam (1 and 2), Seosan, Pyeongchang, Hoengseong, Geumsan, and Goesan (Jo et al., 2016; Kim et al., 2016; Lail et al., 2017). Fresh clubroot galls were collected and washed for isolation of P. brassicae genomic DNA.
DNA was isolated following the method of Laila et al. (2017). Galls from all isolates were cleaned in running tap water and the outer surface was peeled off. The cleaned galls were then surface-sterilized with 70% (v/v) ethanol for 60 s. Approximately 100 mg of gall tissue was collected in a 1.5 ml Eppendorf tube. Individual gall samples were homogenized in 1.5 ml cetrimethyl ammonium bromide (CTAB) (20 g CTAB, 12.11 g Tris, 81.8 g NaCl, and 7.4 g·l−1 disodium-ethylene diamine tetraacetic acid (EDTA); pH 8.0) solution with 30 μl proteinase K (20 mg·l−1, GeneScan) and 30 μl DNaseI (Sigma, St. Louis, MO, USA). The homogenized galls were then incubated at 60°C for 2 h. DNaseI eliminates Brassica rapa host DNA as much as possible (Buhariwalla and Mithen, 1995; Buhariwalla et al., 1995; Manzanares-Dauleux et al., 2001) as it is believed that DNA of P. brassicae become integrated with host DNA during infection process (Bryngelsson et al., 1988). The homogenized suspensions were then centrifuged at 10,000 × g for 5 min. SFG1 Buffer (400 μl) and RNase A (4 μl, Omega Bio-Tek) were mixed with the supernatant in a 1.5 ml tube and vortexed at a maximum speed. The EZNA SP Fungal DNA Miniprep Kit (Omega Bio-Tek, Doraville, GA, USA) was used to extract P. brassicae DNA, following the manufacturer’s protocol. DNA was stored at −20°C after its quantity and quality were assessed with a Nanodrop ND-1000 spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA).
A Japanese cabbage variety (Akimeki) that is resistant to clubroot disease was used as a control to assess the specificity of P. brassicae host genomic DNA. Host genomic DNA was extracted with the Qiagen DNeasy Minikit following the manufacturer’s instructions (Qiagen, Hilden, Germany).
The Phusion High-Fidelity DNA PCR kit (New England Biolabs, Inc., Ipswich, MA, USA) was used to amplify genomic DNA of 11 P. brassicae isolates. Each 50 μl PCR reaction included 1 μl gDNA, 2 μl of each primer, 0.6 μl Phusion DNA Polymerase, 10 μl Phusion HF Buffer, 4 μL dNTPs, and 29.4 μl sterile distilled water (see Table 2 for primer sequences). Reactions were pre-incubated for 5 min at 95°C followed by 35 cycles of amplification at 95°C for 30 s, 58°C for 30 s and 72°C for 45 s in an Eppendorf thermal cycler (Eppendorf AG, Hamburg, Germany). The melting of the mixture was carried out at 72°C for 5 min. PCR products were visualized on a 1.2% agarose gel with 1 × TBE buffer.
By comparing genomic DNA sequences, we have found several regions that vary among P. brassicae isolates. There e3 and ZJ-1 isolates had 59.2% and 59.0% GC content, respectively. Notable variation in %GC content was also evident in four Korean P. brassicae isolates, ranging from 56.0-59.0 (Fig. 1). We used the e3 isolate as a reference genome for the purpose of genome comparisons.
Out of 117 designed markers, 20 primers were e3-specific, 61 primers were ZJ1-specific, two primers were Daejeon-specific, 17 primers were Yeoncheon-specific, six primers were Haenam2-specific, and 11 primers were Gangneung1-specific. Only 12 markers amplified genomic DNA of either a single isolate or two isolates of the same race. The remaining 107 marker sets were non-specific (Supplementary Fig. 1). Two Yeoncheon-specific primers, Ycheon9 and Ycheon10, produced only a single amplicon (Fig. 2), indicating that these sequences characterized amplified region (SCAR) markers might enable specific detection of the Yeoncheon isolate. The primer pair Ycheon14 amplified the targeted region in both the Yeoncheon and Hoengseong isolates (Fig. 2). Since both of these isolates belong to race 2, this marker may be broadly useful in detecting race 2 P. brassicae isolates. The difference in isolate specificity of markers Ycheon9 and Ycheon10 within race 2 might be due to sequence variation between the isolates Yeoncheon and Hoengseong.
Three primer pairs that were designed based on the genomic sequence of the Chinese isolate ZJI (ZJI 3, ZJI 4 and ZJI 5) produced a single amplicon from the Haenam2 isolate (Fig. 3). These three SCAR markers failed to amplify the target region in the other 10 isolates and the control host plant (Fig. 3). It is possible that these markers also amplify the Chinese P. brassicae isolate ZJI, but we did not test this possibility.
Four primer pairs (ZJI 35, ZJI 40, ZJI 41 and ZJI 49) amplified the target region in the Hoengseong isolate only (Fig. 4), indicating that these markers can specifically detect this isolate. Amplification of the Hoengseong isolate with ZJI-specific SCAR markers indicates that the specific primer sequence used is absent in the other P. brassicae isolates examined in this study.
Two other markers (ZJ1 56 and ZJ1 61) amplified the target region in the Pyeongchang-isolate only (Fig. 5). The amplicons were of expected size. These two SCAR markers can be utilized in identifying the Korean P. brassicae isolate Pyeongchang. This result further indicated that ZJ1 isolate has genome similarity with only Pyeongchang-isolate at the primer binding site.
In this study, we developed 12 SCAR markers that identify particular Korean isolates of the clubroot pathogen P. brassicae. Eleven of these markers were specific to four single isolates. Importantly, two Yeoncheon isolate-specific markers perfectly amplified only Yeoncheon isolate of Korea. These markers have the potential to specifically identify Yeoncheon-like isolates from Korean fields. Similarly, the other nine SCAR markers were specific to the Haenam2, Hoengseong, and Pyeongchang isolates. Because three Haenam2-specific markers, four Hoengseong-specific markers, and two Pyeongchang-specific markers were designed based on the genome sequence of the Chinese isolate ZJ1, we predict that these markers may also amplify this Chinese isolate, but we did not test this possibility. We therefore recommended that these markers be tested on all P. brassicae isolates worldwide to further test their specificity and potentially establish them as universal markers. One of the pre-existing limitation at the commencement of this study was lack of any race-specific and isolate-specific markers that can detect a particular P. brassicae isolate. The newly developed marker can efficiently detect four Korean P. brassicae and possibly one Chinese isolate based on genomic DNA sequence variation. Efforts are needed to develop markers to enable detect other Korean P. brassicae isolates.
The authors thank Dr. Gyung Ja Choi, Center for Ecofriendly New Materials, Korea Research Institute of Chemical Technology, Daejeon 34114, Korea for providing P. brassicae isolates. This study was supported by the Center for Horticultural Seed Development (Golden Seed Project No. 213007-05-2-WT611) of the Ministry of Agriculture, Food and Rural Affairs in the Republic of Korea (MAFRA).
Notes
Author contributions
I-SN, J-IP and SKN conceived and designed the study. SKN conducted the in silico analysis. RL and JY prepared fungal spores. RL isolated P. brasicae DNA. JY and HTK conducted PCR amplification. AHKR wrote the manuscript. AHKR, SKN and JY created the tables and figures. All authors read and approved the final draft of the manuscript.
Fig. 1
Alignment of whole genome sequences of Plasmodiophora brassicae. (A) Alignment of five isolates: the dotted rectangles in Yeoncheon and ZL1 indicate the regions where isolate-specific markers were targeted. (B) Identified Yeoncheon- and ZJ1-specific markers regions are highlighted by black arrows.
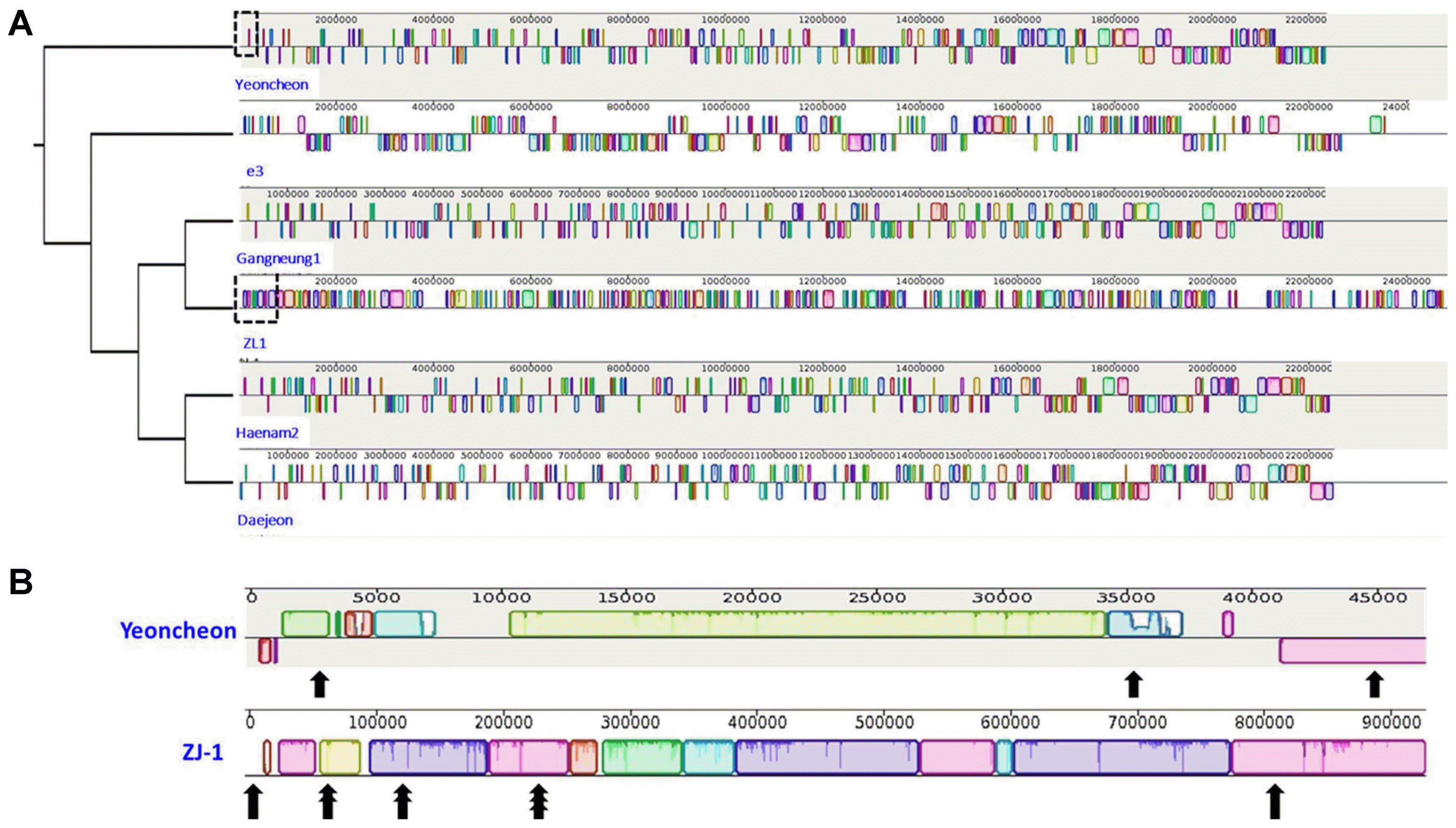
Fig. 2
Three markers specific to the Yeocheon (Ycheon 9 and Ycheon 10), Yeocheon and Hoengseong (Ycheon14) isolates of race 2. Akimeki is a control host. R, race; P, pathotype.

Fig. 3
Three markers showing specificity to the Haenam2 isolate of race 4 and pathotype 4. Akimeki, a Japanese Chinese cabbage cultivar, was a control host. R, race; P, pathotype.

Fig. 4
Four markers showing specificity to the Hoengseong isolate of race 2 and pathotype 1. Akimeki, a Japanese Chinese cabbage cultivar, was a control host. R, race; P, pathotype.

Fig. 5
Four markers showing specificity to the Pyeongchang isolate of race 4 and pathotype 3. Akimeki, a Japanese Chinese cabbage cultivar, was a control host. R, race; P, pathotype.

Table 1
Plasmodiophora brassicae isolates used for comparing genome sequences. Sequences of e3 and ZJ-1 isolates were compared to four Korean isolates
| Isolate | Sequence | Race | Country | Genome size | Reference |
|---|---|---|---|---|---|
| e3 | Genome | Race unknown | Sweden | 24.0508 Mb | Schwelm et al. (2015) |
| ZJ-1 | Genome | Race 1 | China | 24.8311 Mb | Bi et al. (2016) |
| Daejeon | Whole genome re-seq | Race 5 | Korea | 21 Mb | |
| Gangneung 1 | Whole genome re-seq | Race 9 | Korea | 21 Mb | SRA accession: SRP150814 |
| Haenam-2 | Whole genome re-seq | Race 4 | Korea | 21 Mb | |
| Yeoncheon | Whole genome re-seq | Race 2 | Korea | 21 Mb |
Table 2
Forward and reverse primer sequences used to identify Korean Plasmodiophra brassicae isolates
References
Bryngelsson, T, Gustafsson, M, Gréen, B and Lind, C 1988. Uptake of host DNA by the parasitic fungus Plasmodiophora brassicae. Physiol Mol Plant Pathol. 33:163-171.

Buczacki, ST, Toxopeus, H, Mattusch, P, Johnston, TD, Dixon, GR and Hobolth, LA 1975. Study of physiologic specialization in Plasmodiophora brassicae: Proposals for attempted rationalization through an international approach. Trans Br Mycol Soc. 65:295-303.

Buhariwalla, H and Mithen, R 1995. Cloning of a Brassica repetitive DNA element from resting spores of Plasmodiophora brassicae. Physiol Mol Plant Pathol. 47:95-101.

Buhariwalla, H, Greaves, S, Magrath, R and Mithen, R 1995. Development of specific PCR primers for the amplification of polymorphic DNA from the obligate root pathogenPlasmodiophora brassicae. Physiol Mol Plant Pathol. 47:83-94.

Darling, AE, Mau, B and Perna, NT 2010. progressiveMauve: multiple genome alignment with gene gain, loss and rearrangement. PloS One. 5:e11147



Dixon, GR 2009. The occurrence and economic impact of Plasmodiophora brassicae and clubroot disease. J Plant Growth Regul. 28:194-202.

Faggian, R and Strelkov, SE 2009. Detection and measurement of Plasmodiophora brassicae. J Plant Growth Regul. 28:282-288.

Hatakeyama, K, Fujimura, M, Ishida, M and Suzuki, T 2004. New classification method for Plasmodiophora brassicae field isolates in Japan based on resistance of F1 cultivars of Chinese cabbage (Brassica rapa L.) to clubroot. Breed Sci. 54:197-201.

Ito, S, Maehara, T, Tanaka, S, Kameyaiwaki, M, Yano, S and Kishi, F 1997. Cloning of a single-copy DNA sequence unique to Plasmodiophora brassicae. Physiol Mol Plant Pathol. 50:289-300.

Ito, S, Maehara, T, Maruno, E, Tanaka, S, Kameya-Iwaki, M and Kishi, F 2008. Development of a PCR-based assay for the detection of Plasmodiophora brassicae in soil. J Phytopathol. 147:83-88.

Jo, E, Jang, K, Choi, Y, Ahn, K and Choi, G 2016. Resistance of cabbage plants to isolates of Plasmodiophora brassicae. Korean J Hortic Sci Technol. 34:442-452.

Jo, SJ, Shim, SA, Jang, KS, Choi, YH, Kim, JC and Choi, GJ 2011. Resistance of cultivars of Chinese cabbage to Plasmodiophora brassicae isolates of several races collected in Korea. Korean J Hortic Sci Technol. 29:610-616.
Karling, JS 1968. The Plasmodiophorales. 2nd ed. Hafner Publishing Company, New York, USA. 256.
Kearse, M, Moir, R, Wilson, A, Stones-Havas, S, Cheung, M, Sturrock, S, Buxton, S, Cooper, A, Markowitz, S, Duran, C, Thierer, T, Ashton, B, Mentjies, P and Drummond, A 2012. Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics. 28:1647-1649.



Koressaar, T and Remm, M 2007. Enhancements and modifications of primer design program Primer3. Bioinformatics. 23:1289-1291.


Kim, H, Jo, EJ, Choi, YH, Jang, KS and Choi, GJ 2016. Pathotype classification of Plasmodiophora brassicae isolates using clubroot-resistant cultivars of Chinese cabbage. Plant Pathol J. 32:423-430.



Kim, WG, Moon, MH, Kim, JH, Choi, HW and Hong, SK 2009. Occurrence of clubroot on pak-choi caused by Plasmodiophora brassicae. Mycobiology. 37:69-71.



Kuginuki, Y, Yoshikawa, H and Hirai, M 1999. Variation in virulence of Plasmodiophora brassicae in Japan tested with club root resistant cultivars of Chinese cabbage (Brassica rapa L. ssp. pekinensis). Eur J Plant Pathol. 105:327-332.
Laila, R 2018. Molecular investigations towards clubroot resistance and expression of wax biosynthesis genes in Brassicaceae family. Ph D thesis. Sunchon National University, Sunchon, Korea.
Laila, R, Robin, AH, Yang, K, Choi, GJ, Park, JI and Nou, IS 2017. Detection of ribosomal DNA sequence polymorphisms in the protist Plasmodiophora brassicae for the identification of geographical isolates. Int J Mol Sci. 18:84



Li, Y, Yuan, Y, Zhao, Y, Wei, X, Yao, Q, Jiang, W, Wang, Z, Yang, S, Zhang, X and Tian, B 2017. Pathogen identification of clubroot disease in Chinese cabbage from Yuanyang county, Henan province. Mol Plant Breed. 8:45-51.

Li, Z, Niu, YZ, Wu, YC and Guo, SX 2013 Research advances in clubroot disease. Southwest China J Agri Sci. 26:1733-1737 (in Chinese).
Manzanares-Dauleux, MJ, Divaret, I, Baron, F and Thomas, G 2001. Assessment of biological and molecular variability between and within field isolates of Plasmodiophora brassicae. Plant Pathol. 50:165-173.

Nikolaev, SI, Berney, C, Fahrni, JF, Bolivar, I, Polet, S, Mylnikov, AP, Aleshin, VV, Petrov, NB and Pawlowski, J 2004. The twilight of heliozoa and rise of Rhizaria, an emerging supergroup of amoeboid eukaryotes. Proc Natl Acad Sci USA. 101:8066-8071.



Robin, AH, Yi, GE, Laila, R, Yang, K, Park, JI, Kim, HR and Nou, IS 2016. Expression profiling of glucosinolate biosynthetic genes in Brassica oleracea L. var. capitata inbred lines reveals their association with glucosinolate content. Molecules. 21:E787

Schwelm, A, Fogelqvist, J, Knaust, A, Jülke, S, Lilja, T, Bonilla-Rosso, G, Karlsson, M, Shevchenko, A, Dhandapani, V, Choi, SR, Kim, HG, Park, JY, Lim, YP, Ludwig-Müller, J and Dixelius, C 2015. The Plasmodiophora brassicae genome reveals insights in its life cycle and ancestry of chitin synthases. Sci Rep. 5:11153



Somé, A, Manzanares, MJ, Laurens, F, Baron, F, Thomas, G and Rouxel, F 1996. Variation for virulence on Brassica napus L. amongst Plasmodiophora brassicae collections from France and derived single-spore isolates. Plant Pathol. 45:432-439.
Tsushima, S 2000 Perspective of integrated pest management-a case study: clubroot disease of crucifers. J Pest Sci. 25:296-299 (in Japanese).
Untergasser, A, Cutcutache, I, Koressaar, T, Ye, J, Faircloth, BC, Remm, M and Rozen, SG 2012. Primer3-new capabilities and interfaces. Nucleic Acids Res. 40:e115



Voorrips, RE 1995. Plasmodiophora brassicae: aspects of pathogenesis and resistance in Brassica oleracea. Euphytica. 83:139-146.

Wallenhammar, AC 1996. Prevalence of Plasmodiophora brassicae in a spring oilseed rape growing area in central Sweden and factors influencing soil infestation levels. Plant Pathol. 45:710-719.

Wallenhammar, AC and Arwidsson, O 2001. Detection of Plasmodiophora Brassicae by PCR in naturally infested soils. Eur J Plant Pathol. 107:313-321.
Wang, J, Huang, Y, Hu, XL, Niu, YZ, Li, XL and Liang, Y 2008 Study on symptom, yield loss of clubroot and modality of Plasmodiophora brassicae in rape. Chinese J Oil Crop Sci. 30:112-115 (in Chinese).
Williams, PH 1966. A system for determination of races of Plasmodiophora brassicae that infect Cabbage and Rutabaga. Phytopathology. 56:624-626.
Yang, PW, Yang, QZ, Wang, Q, Li, JR and Zeng, L 2002. PCR detection of Plasmodiophora brassicae causing cruciferae clubroot. J Yunnan Agric Univ. 17:137-139.



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Supplement
Supplement Print
Print




