 |
 |
| Plant Pathol J > Volume 35(4); 2019 > Article |
Abstract
Acknowledgments
Fig. 1

Fig. 2
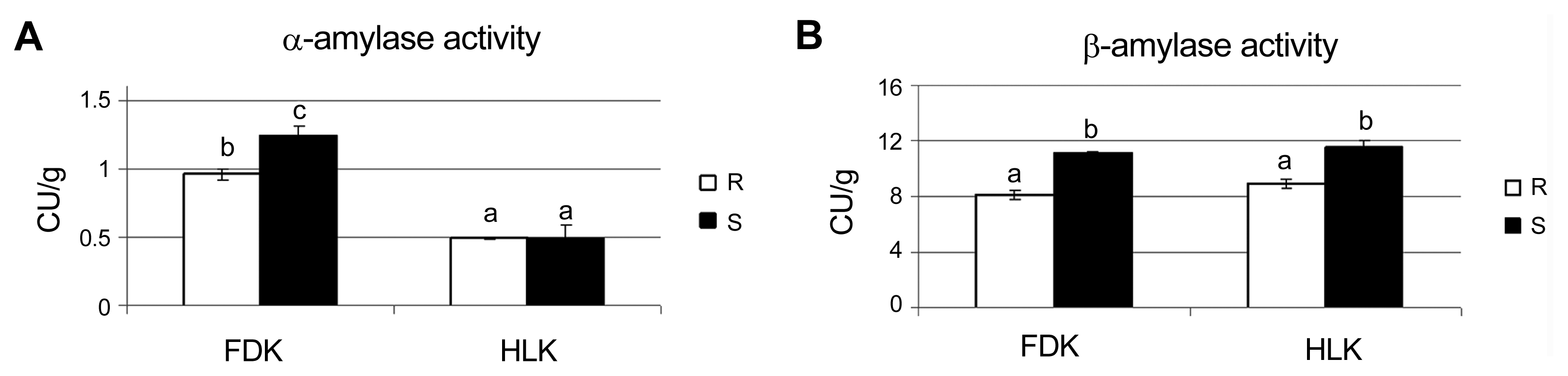
Table 1
FHBi - Fusarium head blight index, FDK - Fusarium-damaged kernels, R - more resistant line, S - more susceptible line; mean values and standard deviations of each parameter calculated after inoculation (three plots) and data from one plot calculated for the control conditions, are shown. Values marked with the same letter did not differ at a significance level of 0.05 (Fisherâs LSD-test).
Table 2
| Rye lines | Conditions after inoculation | Control conditions |
|---|---|---|
| R (TUR) | 9.59668 (Âą1.12) b | 0.735388 a |
| S (HORYZO) | 11.09539 (Âą1.07) b | 0.781604 a |
R - more resistant line, S - more susceptible line; mean values and standard errors of each parameter calculated after inoculation (three plots) and data from one plot calculated for the control conditions, are shown. Values marked with the same letter did not differ at a significance level of 0.05 (Fisherâs LSD-test).
Table 3
| Spot no.1 | Accession2 | Identified protein3 | Score4 | Coverage (%)5 | No. of peptide matched | Molecular function6 |
|---|---|---|---|---|---|---|
| 1 | BAK03012 |
predicted protein [Hordeum vulgare subsp. vulgare] blastp: not identified |
338 | 15 | 5 | - |
| 2 | XP_020150703 | pyrophosphate--fructose 6-phosphate 1-phosphotransferase, subunit beta-like [Aegilops tauschii subsp. tauschii] | 1117 | 29 | 15 | carbohydrate metabolism |
| 3 | BAJ85279 |
predicted protein [Hordeum vulgare subsp. vulgare] blastp: enolase [Aegilops tauschii subsp. tauschii] XP_020194742 |
1493 | 48 | 17 | carbohydrate metabolism |
| 4 | ANW11921 | glyceraldehyde-3-phosphate dehydrogenase [Triticum aestivum] | 745 | 34 | 12 | carbohydrate metabolism |
| 5 | ACN59484 | serpin 2 [Triticum aestivum] | 403 | 13 | 6 | protease inhibition (resistance) |
| 6 | ACO44683 | fructose-bisphosphate aldolase [Secale cereale] | 900 | 38 | 12 | carbohydrate metabolism |
| 7 | EMS58841 | fructose-bisphosphate aldolase [Triticum urartu] | 806 | 35 | 10 | carbohydrate metabolism |
| 8 | ACQ57333 | cytosolic malate dehydrogenase [Triticum aestivum] | 1528 | 61 | 17 | carbohydrate metabolism |
| 9 | BAD06321 | putative caffeoyl CoA O-methyltransferase [Triticum aestivum] | 375 | 22 | 6 | lignin biosynthesis (resistance) |
| 10 | XP_020172398 | 1-Cys peroxiredoxin PER1 [Aegilops tauschii subsp. tauschii] | 376 | 22 | 6 | redox homeostasis |
| 11 | P46226 | triosephosphate isomerase [Secale cereale] | 1071 | 52 | 11 | carbohydrate metabolism |
| 12 | BAK03707 |
predicted protein [Hordeum vulgare subsp. vulgare] blastp: uncharacterized protein |
318 | 42 | 4 | - |
| 13 | - | not identified | - | - | - | - |
| 14 | P12810 | 16.9 kDa class I heat shock protein 1 [Triticum aestivum] | 821 | 62 | 8 | stress response |
| 15 | Q02400 | late embryogenesis abundant protein B19.3 [Hordeum vulgare subsp. vulgare] | 234 | 27 | 4 | stress response |
| 16 | AAZ67071 | cereal-type amylase inhibitor [Secale cereale] | 328 | 43 | 4 | carbohydrate metabolism |
| 17 | - | not identified | - | - | - | - |
| 18 | EMS46390 | serpin-Z2B [Triticum urartu] | 447 | 16 | 7 | protease inhibition (resistance) |
1 Spot numbering was the same as in Fig. 1 and Supplementary Fig. 1.
5 Amino acid sequence coverage for the identified proteins; amino acid sequences for the proteins were shown in Supplementary Fig. 2.
6 Molecular function (www.uniprot.org).
References
- TOOLS
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 2,316 View
- 16 Download
- Related articles
-
Investigation of Genetic Diversity of
Fusarium oxysporum f. sp.fragariae Using PCR-RFLP2017 ;33(2)



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Supplement
Supplement Print
Print



